Matrix metalloproteinase 9 contributes to gut microbe homeostasis in a model of infectious colitis
- PMID: 22694805
- PMCID: PMC3676156
- DOI: 10.1186/1471-2180-12-105
Matrix metalloproteinase 9 contributes to gut microbe homeostasis in a model of infectious colitis
Abstract
Background: Inflammatory bowel diseases are associated with increased expression of zinc-dependent Matrix Metalloproteinase 9 (MMP-9). A stark dysregulation of intestinal mucosal homeostasis has been observed in patients with chronic inflammatory bowel diseases. We therefore sought to determine the contribution of MMP-9 to the pathogenesis of Citrobacter rodentium-induced colitis and its effects on gut microbiome homeostasis.
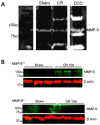
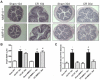
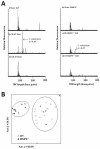
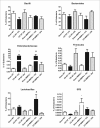
Results: Wild-type and MMP-9-/- mice aged 5-6 weeks were challenged with C. rodentium by orogastric gavage and sacrificed either 10 or 30 days post-infection. Disease severity was assessed by histological analysis of colonic epithelial hyperplasia and by using an in vivo intestinal permeability assay. Changes in the inflammatory responses were measured by using qPCR, and the composition of the fecal microbiome evaluated with both qPCR and terminal restriction fragment length polymorphism. Activation and localization of MMP-9 to the apical surface of the colonic epithelium in response to C. rodentium infection was demonstrated by both zymography and immunocytochemistry. The pro-inflammatory response to infection, including colonic epithelial cell hyperplasia and barrier dysfunction, was similar, irrespective of genotype. Nonmetric multidimensional scaling of terminal restriction fragments revealed a different fecal microbiome composition and C. rodentium colonization pattern between genotypes, with MMP-9-/- having elevated levels of protective segmented filamentous bacteria and interleukin-17, and lower levels of C. rodentium. MMP-9-/- but not wild-type mice were also protected from reductions in fecal microbial diversity in response to the bacterial enteric infection.
Conclusions: These results demonstrate that MMP-9 expression in the colon causes alterations in the fecal microbiome and has an impact on the pathogenesis of bacterial-induced colitis in mice.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

