Intrapleural delivery of MSCs attenuates acute lung injury by paracrine/endocrine mechanism
- PMID: 22697354
- PMCID: PMC4118243
- DOI: 10.1111/j.1582-4934.2012.01597.x
Intrapleural delivery of MSCs attenuates acute lung injury by paracrine/endocrine mechanism
Abstract
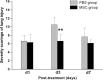
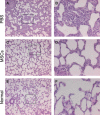
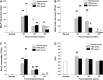
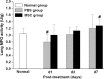
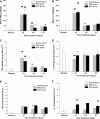
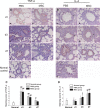
Two different repair mechanisms of mesenchymal stem cells (MSCs) are suggested to participate in the repair of acute lung injury (ALI): (i) Cell engraftment mechanism, (ii) Paracrine/endocrine mechanism. However, the exact roles they play in the repair remain unclear. The aim of the study was to evaluate the role of paracrine/endocrine mechanism using a novel intrapleural delivery method of MSCs. Either 1 × 10(6) MSCs in 300 μl of PBS or 300 μl PBS alone were intrapleurally injected into rats with endotoxin-induced ALI. On days 1, 3 or 7 after injections, samples of lung tissues and bronchoalveolar lavage fluid (BALF) were collected from each rat for assessment of lung injury, biochemical analysis and histology. The distribution of MSCs was also traced by labelling the cells with 4',6-diamidino-2-phenylindole dihydrochloride (DAPI). MSCs intrapleural injection significantly improved LPS-induced lung histopathology compared with PBS-treated group at day 3. There was also a significant decrease in total cell counts and protein concentration in BALF at day 7 in the MSCs -treated rats compared to PBS control group. Tracking the DAPI-marked MSCs showed that there were no exotic MSCs in the lung parenchyma. MSCs administration resulted in a down-regulation of pro-inflammatory response to endotoxin by reducing TNF-α both in the BALF and in the lung, while up-regulating the anti-inflammatory cytokine IL-10 in the lung. In conclusion, treatment with intrapleural MSCs administration markedly attenuates the severity of endotoxin-induced ALI. This role is mediated by paracrine/endocrine repair mechanism of MSCs rather than by the cell engraftment mechanism.
© 2012 The Authors Journal of Cellular and Molecular Medicine © 2012 Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures



Similar articles
-
Angiopoietin-1 Modified Human Umbilical Cord Mesenchymal Stem Cell Therapy for Endotoxin-Induced Acute Lung Injury in Rats.Yonsei Med J. 2017 Jan;58(1):206-216. doi: 10.3349/ymj.2017.58.1.206. Yonsei Med J. 2017. PMID: 27873515 Free PMC article.
-
Bone marrow-derived mesenchymal stem cells protect rats from endotoxin-induced acute lung injury.Chin Med J (Engl). 2011 Sep;124(17):2715-22. Chin Med J (Engl). 2011. PMID: 22040430
-
Therapeutic effect of intravenous bone marrow-derived mesenchymal stem cell transplantation on early-stage LPS-induced acute lung injury in mice.Nan Fang Yi Ke Da Xue Xue Bao. 2012 Mar;32(3):283-90. Nan Fang Yi Ke Da Xue Xue Bao. 2012. PMID: 22445968
-
Human Umbilical Cord Mesenchymal Stem Cells Promote Macrophage PD-L1 Expression and Attenuate Acute Lung Injury in Mice.Curr Stem Cell Res Ther. 2022;17(6):564-575. doi: 10.2174/1574888X17666220127110332. Curr Stem Cell Res Ther. 2022. PMID: 35086457
-
Concise review: Mesenchymal stem cells for acute lung injury: role of paracrine soluble factors.Stem Cells. 2011 Jun;29(6):913-9. doi: 10.1002/stem.643. Stem Cells. 2011. PMID: 21506195 Free PMC article. Review.
Cited by
-
miR-27b represses migration of mouse MSCs to burned margins and prolongs wound repair through silencing SDF-1a.PLoS One. 2013 Jul 22;8(7):e68972. doi: 10.1371/journal.pone.0068972. Print 2013. PLoS One. 2013. PMID: 23894385 Free PMC article.
-
Exosomes derived from human menstrual blood-derived stem cells alleviate fulminant hepatic failure.Stem Cell Res Ther. 2017 Jan 23;8(1):9. doi: 10.1186/s13287-016-0453-6. Stem Cell Res Ther. 2017. PMID: 28115012 Free PMC article.
-
Mesenchymal Stromal Cells Attenuate Infection-Induced Acute Respiratory Distress Syndrome in Animal Experiments: A Meta-Analysis.Cell Transplant. 2020 Jan-Dec;29:963689720969186. doi: 10.1177/0963689720969186. Cell Transplant. 2020. PMID: 33164559 Free PMC article.
-
Exosomes Could Offer New Options to Combat the Long-Term Complications Inflicted by Gestational Diabetes Mellitus.Cells. 2020 Mar 10;9(3):675. doi: 10.3390/cells9030675. Cells. 2020. PMID: 32164322 Free PMC article. Review.
-
Efficacy of Mesenchymal Stromal Cell Therapy for Acute Lung Injury in Preclinical Animal Models: A Systematic Review.PLoS One. 2016 Jan 28;11(1):e0147170. doi: 10.1371/journal.pone.0147170. eCollection 2016. PLoS One. 2016. PMID: 26821255 Free PMC article.
References
-
- Rubenfeld GD, Caldwell E, Peabody E, et al. Incidence and outcomes of acute lung injury. N Engl J Med. 2005;353:1685–93. - PubMed
-
- Zambon M, Vincent JL. Mortality rates for patients with acute lung injury/ARDS have decreased over time. Chest. 2008;133:1120–7. - PubMed
-
- Matthay MA. Treatment of acute lung injury: clinical and experimental studies. Proc Am Thorac Soc. 2008;5:297–9. - PubMed
-
- Xu J, Qu J, Cao L, et al. Mesenchymal stem cell-based angiopoietin-1 gene therapy for acute lung injury induced by lipopolysaccharide in mice. J Pathol. 2008;214:472–81. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

