Dissociation of antibacterial activity and aminoglycoside ototoxicity in the 4-monosubstituted 2-deoxystreptamine apramycin
- PMID: 22699498
- PMCID: PMC3390888
- DOI: 10.1073/pnas.1204073109
Dissociation of antibacterial activity and aminoglycoside ototoxicity in the 4-monosubstituted 2-deoxystreptamine apramycin
Abstract
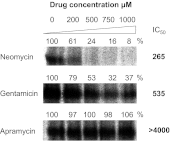
Aminoglycosides are potent antibacterials, but therapy is compromised by substantial toxicity causing, in particular, irreversible hearing loss. Aminoglycoside ototoxicity occurs both in a sporadic dose-dependent and in a genetically predisposed fashion. We recently have developed a mechanistic concept that postulates a key role for the mitochondrial ribosome (mitoribosome) in aminoglycoside ototoxicity. We now report on the surprising finding that apramycin, a structurally unique aminoglycoside licensed for veterinary use, shows little activity toward eukaryotic ribosomes, including hybrid ribosomes which were genetically engineered to carry the mitoribosomal aminoglycoside-susceptibility A1555G allele. In ex vivo cultures of cochlear explants and in the in vivo guinea pig model of chronic ototoxicity, apramycin causes only little hair cell damage and hearing loss but it is a potent antibacterial with good activity against a range of clinical pathogens, including multidrug-resistant Mycobacterium tuberculosis. These data provide proof of concept that antibacterial activity can be dissected from aminoglycoside ototoxicity. Together with 3D structures of apramycin-ribosome complexes at 3.5-Å resolution, our results provide a conceptual framework for further development of less toxic aminoglycosides by hypothesis-driven chemical synthesis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Gale EF, Cundliffe E, Reynolds PE, Richmond MH, Waring MJ. The Molecular Basis of Antibiotic Action. 2nd Ed. London, John Wiley and Sons; 1981.
-
- Chambers HF. Goodman and Gilman's The Pharmaceutical Basis of Therapeutics. 10th Ed. New York: McGraw-Hill; 1996. Chemotherapy of microbial diseases.
-
- Moazed D, Noller HF. Interaction of antibiotics with functional sites in 16S ribosomal RNA. Nature. 1987;327:389–394. - PubMed
-
- Davies JL, Gorini L, Davis BD. Misreading of RNA codewords induced by aminoglycoside antibiotics. Mol Pharmacol. 1965;1:93–106. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

