Cyclotides associate with leaf vasculature and are the products of a novel precursor in petunia (Solanaceae)
- PMID: 22700981
- PMCID: PMC3411041
- DOI: 10.1074/jbc.M112.370841
Cyclotides associate with leaf vasculature and are the products of a novel precursor in petunia (Solanaceae)
Abstract
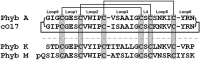
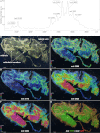
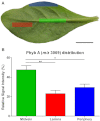
Cyclotides are a large family of plant peptides that are structurally defined by their cyclic backbone and a trifecta of disulfide bonds, collectively known as the cyclic cystine knot (CCK) motif. Structurally similar cyclotides have been isolated from plants within the Rubiaceae, Violaceae, and Fabaceae families and share the CCK motif with trypsin-inhibitory knottins from a plant in the Cucurbitaceae family. Cyclotides have previously been reported to be encoded by dedicated genes or as a domain within a knottin-encoding PA1-albumin-like gene. Here we report the discovery of cyclotides and related non-cyclic peptides we called "acyclotides" from petunia of the agronomically important Solanaceae plant family. Transcripts for petunia cyclotides and acyclotides encode the shortest known cyclotide precursors. Despite having a different precursor structure, their sequences suggest that petunia cyclotides mature via the same biosynthetic route as other cyclotides. We assessed the spatial distribution of cyclotides within a petunia leaf section by MALDI imaging and observed that the major cyclotide component Phyb A was non-uniformly distributed. Dissected leaf midvein extracts contained significantly higher concentrations of this cyclotide compared with the lamina and outer margins of leaves. This is the third distinct type of cyclotide precursor, and Solanaceae is the fourth phylogenetically disparate plant family to produce these structurally conserved cyclopeptides, suggesting either convergent evolution upon the CCK structure or movement of cyclotide-encoding sequences within the plant kingdom.
Figures



Similar articles
-
Discovery and characterization of novel cyclotides originated from chimeric precursors consisting of albumin-1 chain a and cyclotide domains in the Fabaceae family.J Biol Chem. 2011 Jul 8;286(27):24275-87. doi: 10.1074/jbc.M111.229922. Epub 2011 May 19. J Biol Chem. 2011. PMID: 21596752 Free PMC article.
-
Discovery of an unusual biosynthetic origin for circular proteins in legumes.Proc Natl Acad Sci U S A. 2011 Jun 21;108(25):10127-32. doi: 10.1073/pnas.1103660108. Epub 2011 May 18. Proc Natl Acad Sci U S A. 2011. PMID: 21593408 Free PMC article.
-
Discovery, structure, function, and applications of cyclotides: circular proteins from plants.J Exp Bot. 2016 Aug;67(16):4801-12. doi: 10.1093/jxb/erw210. Epub 2016 May 23. J Exp Bot. 2016. PMID: 27222514 Review.
-
Discovery of cyclotides in the fabaceae plant family provides new insights into the cyclization, evolution, and distribution of circular proteins.ACS Chem Biol. 2011 Apr 15;6(4):345-55. doi: 10.1021/cb100388j. Epub 2011 Jan 20. ACS Chem Biol. 2011. PMID: 21194241
-
Oxidative folding of the cystine knot motif in cyclotide proteins.Protein Pept Lett. 2005 Feb;12(2):147-52. doi: 10.2174/0929866053005863. Protein Pept Lett. 2005. PMID: 15723640 Review.
Cited by
-
Isolation and characterization of cyclotides from the leaves of Viola odorata L. using peptidomic and bioinformatic approach.3 Biotech. 2021 May;11(5):211. doi: 10.1007/s13205-021-02763-2. Epub 2021 Apr 11. 3 Biotech. 2021. PMID: 33927999 Free PMC article.
-
Plant Antimicrobial Peptides: State of the Art, In Silico Prediction and Perspectives in the Omics Era.Bioinform Biol Insights. 2020 Sep 2;14:1177932220952739. doi: 10.1177/1177932220952739. eCollection 2020. Bioinform Biol Insights. 2020. PMID: 32952397 Free PMC article. Review.
-
The involvement of cyclotides in the heavy metal tolerance of Viola spp.Sci Rep. 2024 Aug 20;14(1):19306. doi: 10.1038/s41598-024-69018-x. Sci Rep. 2024. PMID: 39164283 Free PMC article.
-
The two-step biosynthesis of cyclic peptides from linear precursors in a member of the plant family Caryophyllaceae involves cyclization by a serine protease-like enzyme.J Biol Chem. 2013 May 3;288(18):12500-10. doi: 10.1074/jbc.M112.437947. Epub 2013 Mar 13. J Biol Chem. 2013. PMID: 23486480 Free PMC article.
-
Two-dimensional graphene as a matrix for MALDI imaging mass spectrometry.J Am Soc Mass Spectrom. 2015 Nov;26(11):1963-6. doi: 10.1007/s13361-015-1243-6. Epub 2015 Sep 1. J Am Soc Mass Spectrom. 2015. PMID: 26323616 Free PMC article.
References
-
- Craik D. J., Daly N. L., Bond T., Waine C. (1999) Plant cyclotides. A unique family of cyclic and knotted proteins that defines the cyclic cystine knot structural motif. J. Mol. Biol. 294, 1327–1336 - PubMed
-
- Plan M. R., Saska I., Cagauan A. G., Craik D. J. (2008) Backbone cyclized peptides from plants show molluscicidal activity against the rice pest Pomacea canaliculata (golden apple snail). J. Agric. Food Chem. 56, 5237–5241 - PubMed
-
- Colgrave M. L., Kotze A. C., Huang Y. H., O'Grady J., Simonsen S. M., Craik D. J. (2008) Cyclotides. Natural, circular plant peptides that possess significant activity against gastrointestinal nematode parasites of sheep. Biochemistry 47, 5581–5589 - PubMed
-
- Gran L. (1973) On the effect of a polypeptide isolated from “Kalata-Kalata” (Oldenlandia affinis DC) on the estrogen-dominated uterus. Acta Pharmacol. Toxicol. 33, 400–408 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources

