Lung effects of inhaled corticosteroids in a rhesus monkey model of childhood asthma
- PMID: 22702509
- PMCID: PMC3913647
- DOI: 10.1111/j.1365-2222.2012.04005.x
Lung effects of inhaled corticosteroids in a rhesus monkey model of childhood asthma
Abstract
Background: The risks for infants and young children receiving inhaled corticosteroid (ICS) therapy are largely unknown. Recent clinical studies indicate that ICS therapy in pre-school children with symptoms of asthma result in decreased symptoms without influencing the clinical disease course, but potentially affect postnatal growth and development. The current study employs a primate experimental model to identify the risks posed by ICS therapy.
Objective: To (1) establish whether ICS therapy in developing primate lungs reverses pulmonary pathobiology associated with allergic airway disease (AAD) and (2) define the impact of ICS on postnatal lung growth and development in primates.
Methods: Infant rhesus monkeys were exposed, from 1 through 6 months, to filtered air (FA) with house dust mite allergen and ozone using a protocol that produces AAD (AAD monkeys), or to FA alone (Control monkeys). From three through 6 months, the monkeys were treated daily with ICS (budesonide) or saline.
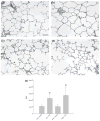
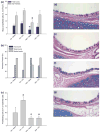
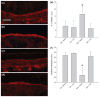
Results: Several AAD manifestations (airflow restrictions, lavage eosinophilia, basement membrane zone thickening, epithelial mucin composition) were reduced with ICS treatment, without adverse effects on body growth or adrenal function; however, airway branching abnormalities and intraepithelial innervation were not reduced. In addition, several indicators of postnatal lung growth and differentiation: vital capacity, inspiratory capacity, compliance, non-parenchymal lung volume and alveolarization, were increased in both AAD and Control monkeys that received ICS treatment.
Conclusions and clinical relevance: Incomplete prevention of pathobiological changes in the airways and disruption of postnatal growth and differentiation of airways and lung parenchyma in response to ICS pose risks for developing primate lungs. These responses also represent two mechanisms that could compromise ICS therapy's ability to alter clinical disease course in young children.
© 2012 Blackwell Publishing Ltd.
Conflict of interest statement
Figures


Similar articles
-
The effects of inhaled corticosteroids on intrinsic responsiveness and histology of airways from infant monkeys exposed to house dust mite allergen and ozone.Toxicol Appl Pharmacol. 2008 Jan 15;226(2):153-60. doi: 10.1016/j.taap.2007.09.005. Epub 2007 Sep 16. Toxicol Appl Pharmacol. 2008. PMID: 17991502
-
[Standard technical specifications for methacholine chloride (Methacholine) bronchial challenge test (2023)].Zhonghua Jie He He Hu Xi Za Zhi. 2024 Feb 12;47(2):101-119. doi: 10.3760/cma.j.cn112147-20231019-00247. Zhonghua Jie He He Hu Xi Za Zhi. 2024. PMID: 38309959 Chinese.
-
Asthma/allergic airways disease: does postnatal exposure to environmental toxicants promote airway pathobiology?Toxicol Pathol. 2007 Jan;35(1):97-110. doi: 10.1080/01926230601132030. Toxicol Pathol. 2007. PMID: 17325978
-
The non-human primate as a model for studying COPD and asthma.Pulm Pharmacol Ther. 2008 Oct;21(5):755-66. doi: 10.1016/j.pupt.2008.01.008. Epub 2008 Feb 1. Pulm Pharmacol Ther. 2008. PMID: 18339566 Review.
-
Inhaled corticosteroids in children with asthma: pharmacologic determinants of safety and efficacy and other clinical considerations.Paediatr Drugs. 2007;9(3):185-94. doi: 10.2165/00148581-200709030-00007. Paediatr Drugs. 2007. PMID: 17523699 Review.
Cited by
-
Nonhuman Primate Models of Respiratory Disease: Past, Present, and Future.ILAR J. 2017 Dec 1;58(2):269-280. doi: 10.1093/ilar/ilx030. ILAR J. 2017. PMID: 29216343 Free PMC article. Review.
-
Use of nonhuman primates in obstructive lung disease research - is it required?Primate Biol. 2017 Jun 30;4(1):131-142. doi: 10.5194/pb-4-131-2017. eCollection 2017. Primate Biol. 2017. PMID: 32110701 Free PMC article. Review.
-
Can early intervention in pediatric asthma improve long-term outcomes? A question that needs an answer.Pediatr Pulmonol. 2019 Mar;54(3):348-357. doi: 10.1002/ppul.24224. Epub 2019 Jan 4. Pediatr Pulmonol. 2019. PMID: 30609252 Free PMC article. Review.
-
Newly divided eosinophils limit ozone-induced airway hyperreactivity in nonsensitized guinea pigs.Am J Physiol Lung Cell Mol Physiol. 2017 Jun 1;312(6):L969-L982. doi: 10.1152/ajplung.00530.2016. Epub 2017 Mar 3. Am J Physiol Lung Cell Mol Physiol. 2017. PMID: 28258108 Free PMC article.
-
Why primate models matter.Am J Primatol. 2014 Sep;76(9):801-27. doi: 10.1002/ajp.22281. Epub 2014 Apr 10. Am J Primatol. 2014. PMID: 24723482 Free PMC article. Review.
References
-
- Sly PD. The early origins of asthma: who is really at risk? Curr Opin Allergy Clin Immunol. 2011;11:24–8. - PubMed
-
- Mortimer KM, Neas LM, Dockery DW, Redline S, Tager IB. The effect of air pollution on inner-city children with asthma. Eur Respir J. 2002;19:699–705. - PubMed
-
- Yunginger JW, Reed CE, O’Connell EJ, Melton LJ, 3rd, O’Fallon WM, Silverstein MD. A community-based study of the epidemiology of asthma. Incidence rates, 1964–1983. Am Rev Respir Dis. 1992;146:888–94. - PubMed
-
- Rosenstreich DL, Eggleston P, Kattan M, et al. The role of cockroach allergy and exposure to cockroach allergen in causing morbidity among inner-city children with asthma. N Engl J Med. 1997;336:1356–63. - PubMed

