Outer-sphere direction in iridium C-H borylation
- PMID: 22703452
- PMCID: PMC3587652
- DOI: 10.1021/ja303443m
Outer-sphere direction in iridium C-H borylation
Abstract
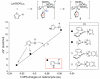
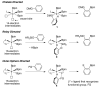
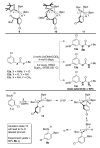
The NHBoc group affords ortho selective C-H borylations in arenes and alkenes. Experimental and computational studies support an outer sphere mechanism where the N-H proton hydrogen bonds to a boryl ligand oxygen. The regioselectivities are unique and complement those of directed ortho metalations.
Figures





References
-
- Mkhalid IAI, Barnard JH, Marder TB, Murphy JM, Hartwig JF. Chem. Rev. 2010;110:890. - PubMed
-
- Boebel TA, Hartwig JF. J. Am. Chem. Soc. 2008;130:7534. - PubMed
- Kawamorita S, Ohmiya H, Hara K, Fukuoka A, Sawamura M. J. Am. Chem. Soc. 2009;131:5058. - PubMed
- Ishiyama T, Isou H, Kikuchi T, Miyaura N. Chem. Commun. 2010;46:159. - PubMed
- Robbins DW, Boebel TA, Hartwig JF. J. Am. Chem. Soc. 2010;132:4068. - PubMed
- Ros A, Estepa B, Lopez-Rodriguez R, Alvarez E, Fernandez R, Lassaletta JM. Angew. Chem., Int. Ed. 2011;50:11724. - PubMed
- Dai H-X, Yu J-Q. J. Am. Chem. Soc. 2012;134:134. - PubMed
- Ros A, Lopez-Rodriguez R, Estepa B, Alvarez E, Fernandez R, Lassaletta JM. J. Am. Chem. Soc. 2012;134:4573. - PubMed
- Xiao B, Li Y-M, Liu Z-J, Yang H-Y, Fu Y. Chem. Commun. 2012;48:4854. - PubMed
-
-
Although “outer-sphere” commonly refers to eletron transfer, it has been used to describe mechanisms for hydrogen transfer and nucleophilic addition to π-allyl Pd compounds: Samec JSM, Backvall JE, Andersson PG, Brandt P. Chem. Soc. Rev. 2006;35:237. Trost BM, Xu JY, Schmidt TJ. Am. Chem. Soc. 2009;131:18343.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

