Dendritic cell populations with different concentrations of lipid regulate tolerance and immunity in mouse and human liver
- PMID: 22705178
- PMCID: PMC3459067
- DOI: 10.1053/j.gastro.2012.06.003
Dendritic cell populations with different concentrations of lipid regulate tolerance and immunity in mouse and human liver
Abstract
Background & aims: Immune cells of the liver must be able to recognize and react to pathogens yet remain tolerant to food molecules and other nonpathogens. Dendritic cells (DCs) are believed to contribute to hepatic tolerance. Lipids have been implicated in dysfunction of DCs in cancer. Therefore, we investigated whether high lipid content in liver DCs affects induction of tolerance.
Methods: Mouse and human hepatic nonparenchymal cells were isolated by mechanical and enzymatic digestion. DCs were purified by fluorescence-activated cell sorting or with immunomagnetic beads. DC lipid content was assessed by flow cytometry, immune fluorescence, and electron microscopy and by measuring intracellular component lipids. DC activation was determined from surface phenotype and cytokine profile. DC function was assessed in T-cell, natural killer (NK) cell, and NKT cell coculture assays as well as in vivo.
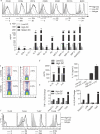
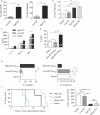
Results: We observed 2 distinct populations of hepatic DCs in mice and humans based on their lipid content and expression of markers associated with adipogenesis and lipid metabolism. This lipid-based dichotomy in DCs was unique to the liver and specific to DCs compared with other hepatic immune cells. However, rather than mediate tolerance, the liver DC population with high concentrations of lipid was immunogenic in multiple models; they activated T cells, NK cells, and NKT cells. Conversely, liver DCs with low levels of lipid induced regulatory T cells, anergy to cancer, and oral tolerance. The immunogenicity of lipid-rich liver DCs required their secretion of tumor necrosis factor α and was directly related to their high lipid content; blocking DC synthesis of fatty acids or inhibiting adipogenesis (by reducing endoplasmic reticular stress) reduced DC immunogenicity.
Conclusions: Human and mouse hepatic DCs are composed of distinct populations that contain different concentrations of lipid, which regulates immunogenic versus tolerogenic responses in the liver.
Copyright © 2012 AGA Institute. Published by Elsevier Inc. All rights reserved.
Figures




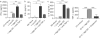
References
-
- Tsuji NM, et al. Oral tolerance: intestinal homeostasis and antigen-specific regulatory T cells. Trends Immunol. 2008;29:532–40. - PubMed
-
- Zavadsky KE, et al. Liver metastases from colorectal carcinoma: incidence, resectability, and survival results. Am Surg. 1994;60:929–33. - PubMed
-
- Limmer A, et al. Cross-presentation of oral antigens by liver sinusoidal endothelial cells leads to CD8 T cell tolerance. Eur J Immunol. 2005;35:2970–81. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

