Structure of nucleophosmin DNA-binding domain and analysis of its complex with a G-quadruplex sequence from the c-MYC promoter
- PMID: 22707729
- PMCID: PMC3410995
- DOI: 10.1074/jbc.M112.371013
Structure of nucleophosmin DNA-binding domain and analysis of its complex with a G-quadruplex sequence from the c-MYC promoter
Abstract
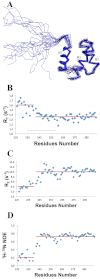
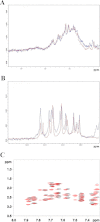
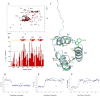
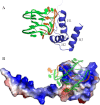
Nucleophosmin (NPM1) is a nucleocytoplasmic shuttling protein, mainly localized at nucleoli, that plays a key role in several cellular functions, including ribosome maturation and export, centrosome duplication, and response to stress stimuli. More than 50 mutations at the terminal exon of the NPM1 gene have been identified so far in acute myeloid leukemia; the mutated proteins are aberrantly and stably localized in the cytoplasm due to high destabilization of the NPM1 C-terminal domain and the appearance of a new nuclear export signal. We have shown previously that the 70-residue NPM1 C-terminal domain (NPM1-C70) is able to bind with high affinity a specific region at the c-MYC gene promoter characterized by parallel G-quadruplex structure. Here we present the solution structure of the NPM1-C70 domain and NMR analysis of its interaction with a c-MYC-derived G-quadruplex. These data were used to calculate an experimentally restrained molecular docking model for the complex. The NPM1-C70 terminal three-helix bundle binds the G-quadruplex DNA at the interface between helices H1 and H2 through electrostatic interactions with the G-quadruplex phosphate backbone. Furthermore, we show that the 17-residue lysine-rich sequence at the N terminus of the three-helix bundle is disordered and, although necessary, does not participate directly in the contact surface in the complex.
Figures

References
-
- Grisendi S., Mecucci C., Falini B., Pandolfi P. P. (2006) Nucleophosmin and cancer. Nat. Rev. Cancer 6, 493–505 - PubMed
-
- Falini B., Sportoletti P., Martelli M. P. (2009) Acute myeloid leukemia with mutated NPM1: diagnosis, prognosis and therapeutic perspectives. Curr. Opin. Oncol. 21, 573–581 - PubMed
-
- Falini B., Gionfriddo I., Cecchetti F., Ballanti S., Pettirossi V., Martelli M. P. (2011) Acute myeloid leukemia with mutated nucleophosmin (NPM1): any hope for a targeted therapy? Blood Rev. 25, 247–254 - PubMed
-
- Falini B., Mecucci C., Tiacci E., Alcalay M., Rosati R., Pasqualucci L., La Starza R., Diverio D., Colombo E., Santucci A., Bigerna B., Pacini R., Pucciarini A., Liso A., Vignetti M., Fazi P., Meani N., Pettirossi V., Saglio G., Mandelli F., Lo-Coco F., Pelicci P. G., Martelli M. F.; GIMEMA Acute Leukemia Working Party (2005) Cytoplasmic nucleophosmin in acute myelogenous leukemia with a normal karyotype. N. Engl. J. Med. 352, 254–266 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources

