Analytical and biological methods for probing the blood-brain barrier
- PMID: 22708905
- PMCID: PMC3744104
- DOI: 10.1146/annurev-anchem-062011-143002
Analytical and biological methods for probing the blood-brain barrier
Abstract
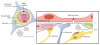
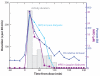
The blood-brain barrier (BBB) is an important interface between the peripheral and central nervous systems. It protects the brain against the infiltration of harmful substances and regulates the permeation of beneficial endogenous substances from the blood into the extracellular fluid of the brain. It can also present a major obstacle in the development of drugs that are targeted for the central nervous system. Several methods have been developed to investigate the transport and metabolism of drugs, peptides, and endogenous compounds at the BBB. In vivo methods include intravenous injection, brain perfusion, positron emission tomography, and microdialysis sampling. Researchers have also developed in vitro cell-culture models that can be employed to investigate transport and metabolism at the BBB without the complication of systemic involvement. All these methods require sensitive and selective analytical methods to monitor the transport and metabolism of the compounds of interest at the BBB.
Figures
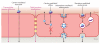


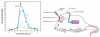



Similar articles
-
Separation methods that are capable of revealing blood-brain barrier permeability.J Chromatogr B Analyt Technol Biomed Life Sci. 2003 Nov 25;797(1-2):241-54. doi: 10.1016/s1570-0232(03)00605-6. J Chromatogr B Analyt Technol Biomed Life Sci. 2003. PMID: 14630153 Review.
-
Microfluidic blood-brain barrier model provides in vivo-like barrier properties for drug permeability screening.Biotechnol Bioeng. 2017 Jan;114(1):184-194. doi: 10.1002/bit.26045. Epub 2016 Jul 21. Biotechnol Bioeng. 2017. PMID: 27399645 Free PMC article.
-
On-line microdialysis coupled to analytical systems.J Chromatogr Sci. 2008 Mar;46(3):276-87. doi: 10.1093/chromsci/46.3.276. J Chromatogr Sci. 2008. PMID: 18334095 Review.
-
[Development of Novel Methodology and Its Application for Clarifying the Transport Function of the Blood-brain Barrier].Yakugaku Zasshi. 2021;141(4):447-462. doi: 10.1248/yakushi.20-00232. Yakugaku Zasshi. 2021. PMID: 33790111 Review. Japanese.
-
Methodological considerations of intracerebral microdialysis in pharmacokinetic studies on drug transport across the blood-brain barrier.Brain Res Brain Res Rev. 1997 Sep 30;25(1):27-49. doi: 10.1016/s0165-0173(97)00014-3. Brain Res Brain Res Rev. 1997. PMID: 9370049 Review.
Cited by
-
Parallelizable Microfluidic Platform to Model and Assess In Vitro Cellular Barriers: Technology and Application to Study the Interaction of 3D Tumor Spheroids with Cellular Barriers.Biosensors (Basel). 2021 Sep 3;11(9):314. doi: 10.3390/bios11090314. Biosensors (Basel). 2021. PMID: 34562904 Free PMC article.
-
3D printed devices with integrated collagen scaffolds for cell culture studies including transepithelial/transendothelial electrical resistance (TEER) measurements.Anal Chim Acta. 2022 Aug 15;1221:340166. doi: 10.1016/j.aca.2022.340166. Epub 2022 Jul 13. Anal Chim Acta. 2022. PMID: 35934386 Free PMC article.
-
Models and methods to evaluate transport of drug delivery systems across cellular barriers.J Vis Exp. 2013 Oct 17;(80):e50638. doi: 10.3791/50638. J Vis Exp. 2013. PMID: 24192611 Free PMC article.
-
Design and Validation of a Human Brain Endothelial Microvessel-on-a-Chip Open Microfluidic Model Enabling Advanced Optical Imaging.Front Bioeng Biotechnol. 2020 Sep 28;8:573775. doi: 10.3389/fbioe.2020.573775. eCollection 2020. Front Bioeng Biotechnol. 2020. PMID: 33117784 Free PMC article.
-
Development of Polymer-Based Nanoformulations for Glioblastoma Brain Cancer Therapy and Diagnosis: An Update.Polymers (Basel). 2021 Nov 26;13(23):4114. doi: 10.3390/polym13234114. Polymers (Basel). 2021. PMID: 34883617 Free PMC article. Review.
References
-
- Cardoso FL, Brites D, Brito MA. Looking at the blood-brain barrier: molecular anatomy and possible investigation approaches. Brain Res. Rev. 2010;64:328–63. - PubMed
-
- Abbott NJ, Roennbaeck L, Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006;7:41–53. - PubMed
-
- Abbott NJ, Patabendige AA, Dolman DE, Yusof SR, Begley DJ. Structure and function of the blood-brain barrier. Neurobiol. Dis. 2010;37:13–25. - PubMed
-
- Jain AJ, SK Drug targeting to brain: a review. Curr. Nanosci. 2011;7:21–36.
-
- Jeffrey P, Summerfield S. Assessment of the blood-brain barrier in CNS drug discovery. Neurobiol. Dis. 2010;37:33–37. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

