Regulation of neuronal bioenergy homeostasis by glutamate
- PMID: 22709672
- PMCID: PMC3430810
- DOI: 10.1016/j.neuint.2012.06.003
Regulation of neuronal bioenergy homeostasis by glutamate
Abstract
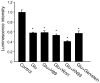
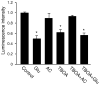
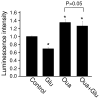
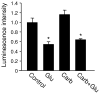
Bioenergy homeostasis is crucial in maintaining normal cell function and survival and it is thus important to understand cellular mechanisms underlying its regulation. Neurons use a large amount of ATP to maintain membrane potential and synaptic communication, making the brain the most energy consuming organ in the body. Glutamate mediates a large majority of synaptic transmission which is responsible for the expression of neural plasticity and higher brain functions. Most of the energy cost is attributable to the glutamatergic system; under pathological conditions such as stroke and brain ischemia, neural energy depletion is accompanied by a massive release of glutamate. However, the specific cellular processes implicated in glutamate-dependent bioenergy dynamics are not well understood. We find that glutamate induces a rapid and dramatic reduction of ATP levels in neurons, through reduced ATP genesis and elevated consumption. ATP reduction depends on NMDA receptor activity, but is not a result of neuronal firing, gap junction-mediated leaking or intracellular signaling. Similar changes in ATP levels are also induced by synaptic glutamate accumulation following suppression of glutamate transporter activity. Furthermore, the glutamate-induced ATP down-regulation is blocked by the sodium pump inhibitor ouabain, suggesting the sodium pump as the primary energy consumer during glutamate stimulation. These data suggest the important role of glutamate in the control of cellular ATP homeostasis.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Conflict of interest statement
The authors state no conflict of interest.
Figures
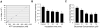
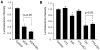
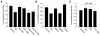
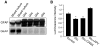
Similar articles
-
ATP released by astrocytes mediates glutamatergic activity-dependent heterosynaptic suppression.Neuron. 2003 Dec 4;40(5):971-82. doi: 10.1016/s0896-6273(03)00717-7. Neuron. 2003. PMID: 14659095
-
Deletion of Neuronal GLT-1 in Mice Reveals Its Role in Synaptic Glutamate Homeostasis and Mitochondrial Function.J Neurosci. 2019 Jun 19;39(25):4847-4863. doi: 10.1523/JNEUROSCI.0894-18.2019. Epub 2019 Mar 29. J Neurosci. 2019. PMID: 30926746 Free PMC article.
-
Glutamate metabolism and recycling at the excitatory synapse in health and neurodegeneration.Neuropharmacology. 2021 Sep 15;196:108719. doi: 10.1016/j.neuropharm.2021.108719. Epub 2021 Jul 15. Neuropharmacology. 2021. PMID: 34273389 Review.
-
Neuron-glia interactions in glutamatergic neurotransmission: roles of oxidative and glycolytic adenosine triphosphate as energy source.J Neurosci Res. 2011 Dec;89(12):1926-34. doi: 10.1002/jnr.22746. Epub 2011 Sep 14. J Neurosci Res. 2011. PMID: 21919035 Review.
-
Excitation-transcription coupling via calcium/calmodulin-dependent protein kinase/ERK1/2 signaling mediates the coordinate induction of VGLUT2 and Narp triggered by a prolonged increase in glutamatergic synaptic activity.J Biol Chem. 2010 May 7;285(19):14366-76. doi: 10.1074/jbc.M109.080069. Epub 2010 Mar 8. J Biol Chem. 2010. PMID: 20212045 Free PMC article.
Cited by
-
Ouabain Reverts CUS-Induced Disruption of the HPA Axis and Avoids Long-Term Spatial Memory Deficits.Biomedicines. 2023 Apr 14;11(4):1177. doi: 10.3390/biomedicines11041177. Biomedicines. 2023. PMID: 37189795 Free PMC article.
-
Cellular basis of learning and memory in the carotid body.Front Synaptic Neurosci. 2022 Aug 15;14:902319. doi: 10.3389/fnsyn.2022.902319. eCollection 2022. Front Synaptic Neurosci. 2022. PMID: 36046221 Free PMC article. Review.
-
A Novel Mechanism of Spine Damages in Stroke via DAPK1 and Tau.Cereb Cortex. 2015 Nov;25(11):4559-71. doi: 10.1093/cercor/bhv096. Epub 2015 May 20. Cereb Cortex. 2015. PMID: 25995053 Free PMC article.
-
The Influence of Na(+), K(+)-ATPase on Glutamate Signaling in Neurodegenerative Diseases and Senescence.Front Physiol. 2016 Jun 2;7:195. doi: 10.3389/fphys.2016.00195. eCollection 2016. Front Physiol. 2016. PMID: 27313535 Free PMC article. Review.
-
Conditioned medium of human adipose-derived mesenchymal stem cells mediates protection in neurons following glutamate excitotoxicity by regulating energy metabolism and GAP-43 expression.Metab Brain Dis. 2014 Mar;29(1):193-205. doi: 10.1007/s11011-014-9490-y. Epub 2014 Jan 25. Metab Brain Dis. 2014. PMID: 24458787 Free PMC article.
References
-
- Anwyl R. Metabotropic glutamate receptor-dependent long-term potentiation. Neuropharmacology. 2009;56:735–740. - PubMed
-
- Attwell D, Laughlin SB. An energy budget for signaling in the grey matter of the brain. J Cereb Blood Flow Metab. 2001;21:1133–1145. - PubMed
-
- Bennett MV, Zukin RS. Electrical coupling and neuronal synchronization in the Mammalian brain. Neuron. 2004;41:495–511. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

