Endogenous production of fibronectin is required for self-renewal of cultured mouse embryonic stem cells
- PMID: 22710062
- PMCID: PMC3582329
- DOI: 10.1016/j.yexcr.2012.06.009
Endogenous production of fibronectin is required for self-renewal of cultured mouse embryonic stem cells
Abstract
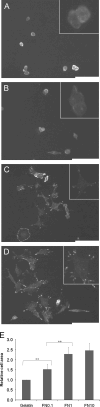
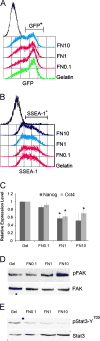
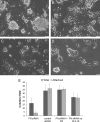
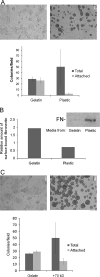
Pluripotent cells are attached to the extracellular matrix (ECM) as they make cell fate decisions within the stem cell niche. Here we show that the ubiquitous ECM protein fibronectin is required for self-renewal decisions by cultured mouse embryonic stem (mES) cells. Undifferentiated mES cells produce fibronectin and assemble a fibrillar matrix. Increasing the level of substrate fibronectin increased cell spreading and integrin receptor signaling through focal adhesion kinase, while concomitantly inducing the loss of Nanog and Oct4 self-renewal markers. Conversely, reducing fibronectin production by mES cells growing on a feeder-free gelatin substrate caused loss of cell adhesion, decreased integrin signaling, and decreased expression of self-renewal markers. These effects were reversed by providing the cells with exogenous fibronectin, thereby restoring adhesion to the gelatin substrate. Interestingly, mES cells do not adhere directly to the gelatin substrate, but rather adhere indirectly through gelatin-bound fibronectin, which facilitates self-renewal via its effects on cell adhesion. These results provide new insights into the mechanism of regulation of self-renewal by growth on a gelatin-coated surface. The effects of increasing or decreasing fibronectin levels show that self-renewal depends on an intermediate level of cell-fibronectin interactions. By providing cell adhesive signals that can act with other self-renewal factors to maintain mES cell pluripotency, fibronectin is therefore a necessary component of the self-renewal signaling pathway in culture.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Integrins regulate mouse embryonic stem cell self-renewal.Stem Cells. 2007 Dec;25(12):3005-15. doi: 10.1634/stemcells.2007-0103. Epub 2007 Aug 23. Stem Cells. 2007. PMID: 17717067
-
Inhibition of Focal Adhesion Kinase Signaling by Integrin α6β1 Supports Human Pluripotent Stem Cell Self-Renewal.Stem Cells. 2016 Jul;34(7):1753-64. doi: 10.1002/stem.2349. Epub 2016 Mar 27. Stem Cells. 2016. PMID: 26930028
-
Fibronectin-mediated upregulation of α5β1 integrin and cell adhesion during differentiation of mouse embryonic stem cells.Cell Adh Migr. 2011 Jan-Feb;5(1):73-82. doi: 10.4161/cam.5.1.13704. Epub 2011 Jan 1. Cell Adh Migr. 2011. PMID: 20962574 Free PMC article.
-
Transforming growth factor-beta superfamily in mouse embryonic stem cell self-renewal.Vitam Horm. 2011;87:341-65. doi: 10.1016/B978-0-12-386015-6.00035-4. Vitam Horm. 2011. PMID: 22127250 Review.
-
Molecular mechanisms involved in self-renewal and pluripotency of embryonic stem cells.J Cell Physiol. 2007 May;211(2):279-86. doi: 10.1002/jcp.20978. J Cell Physiol. 2007. PMID: 17195167 Review.
Cited by
-
Defined extracellular matrix components are necessary for definitive endoderm induction.Stem Cells. 2013 Oct;31(10):2084-94. doi: 10.1002/stem.1453. Stem Cells. 2013. PMID: 23766144 Free PMC article.
-
Layer-Number-Dependent Effects of Graphene Oxide on the Pluripotency of Mouse Embryonic Stem Cells Through the Regulation of the Interaction Between the Extracellular Matrix and Integrins.Int J Nanomedicine. 2021 Jun 4;16:3819-3832. doi: 10.2147/IJN.S301892. eCollection 2021. Int J Nanomedicine. 2021. PMID: 34121840 Free PMC article.
-
Reversible modulation of myofibroblast differentiation in adipose-derived mesenchymal stem cells.PLoS One. 2014 Jan 23;9(1):e86865. doi: 10.1371/journal.pone.0086865. eCollection 2014. PLoS One. 2014. PMID: 24466271 Free PMC article.
-
Jun-Mediated Changes in Cell Adhesion Contribute to Mouse Embryonic Stem Cell Exit from Ground State Pluripotency.Stem Cells. 2016 May;34(5):1213-24. doi: 10.1002/stem.2294. Epub 2016 Feb 13. Stem Cells. 2016. PMID: 26850660 Free PMC article.
-
The self-renewal of mouse embryonic stem cells is regulated by cell-substratum adhesion and cell spreading.Int J Biochem Cell Biol. 2013 Nov;45(11):2698-705. doi: 10.1016/j.biocel.2013.07.001. Epub 2013 Jul 17. Int J Biochem Cell Biol. 2013. PMID: 23871934 Free PMC article.
References
-
- Smith AG. Embryo-derived stem cells: of mice and men. Annu Rev Cell Dev Biol. 2001;17:435–462. - PubMed
-
- Chambers I, Colbya D, Robertsona M, Nicholsa J, Leea S, Tweediea S, Smith A. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell. 2003;113:643–655. - PubMed
-
- Mitsui K, Tokuzawa Y, Itoh H, Segaw K, Murakami M, Takahashi K, Maruyama M, Maeda M, Yamanaka S. The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells. Cell. 2003;113:631–642. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

