Kruppel-associated Box (KRAB)-associated co-repressor (KAP-1) Ser-473 phosphorylation regulates heterochromatin protein 1β (HP1-β) mobilization and DNA repair in heterochromatin
- PMID: 22715096
- PMCID: PMC3431694
- DOI: 10.1074/jbc.M112.368381
Kruppel-associated Box (KRAB)-associated co-repressor (KAP-1) Ser-473 phosphorylation regulates heterochromatin protein 1β (HP1-β) mobilization and DNA repair in heterochromatin
Abstract
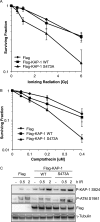
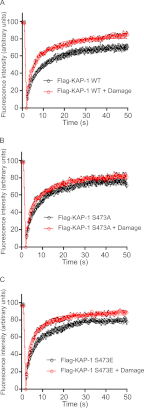
The DNA damage response encompasses a complex series of signaling pathways that function to regulate and facilitate the repair of damaged DNA. Recent studies have shown that the repair of transcriptionally inactive chromatin, named heterochromatin, is dependent upon the phosphorylation of the co-repressor, Krüppel-associated box (KRAB) domain-associated protein (KAP-1), by the ataxia telangiectasia-mutated (ATM) kinase. Co-repressors, such as KAP-1, function to regulate the rigid structure of heterochromatin by recruiting histone-modifying enzymes, such HDAC1/2, SETDB1, and nucleosome-remodeling complexes such as CHD3. Here, we have characterized a phosphorylation site in the HP1-binding domain of KAP-1, Ser-473, which is phosphorylated by the cell cycle checkpoint kinase Chk2. Expression of a nonphosphorylatable S473A mutant conferred cellular sensitivity to DNA-damaging agents and led to defective repair of DNA double-strand breaks in heterochromatin. In addition, cells expressing S473A also displayed defective mobilization of the HP1-β chromodomain protein. The DNA repair defect observed in cells expressing S473A was alleviated by depletion of HP1-β, suggesting that phosphorylation of KAP-1 on Ser-473 promotes the mobilization of HP1-β from heterochromatin and subsequent DNA repair. These results suggest a novel mechanism of KAP-1-mediated chromatin restructuring via Chk2-regulated HP1-β exchange from heterochromatin, promoting DNA repair.
Figures


Similar articles
-
The impact of heterochromatin on DSB repair.Biochem Soc Trans. 2009 Jun;37(Pt 3):569-76. doi: 10.1042/BST0370569. Biochem Soc Trans. 2009. PMID: 19442252
-
The ATM substrate KAP1 controls DNA repair in heterochromatin: regulation by HP1 proteins and serine 473/824 phosphorylation.Mol Cancer Res. 2012 Mar;10(3):401-14. doi: 10.1158/1541-7786.MCR-11-0134. Epub 2011 Dec 28. Mol Cancer Res. 2012. PMID: 22205726 Free PMC article.
-
Roles of Kruppel-associated Box (KRAB)-associated Co-repressor KAP1 Ser-473 Phosphorylation in DNA Damage Response.J Biol Chem. 2012 Jun 1;287(23):18937-52. doi: 10.1074/jbc.M111.313262. Epub 2012 Apr 11. J Biol Chem. 2012. PMID: 22496453 Free PMC article.
-
The influence of heterochromatin on DNA double strand break repair: Getting the strong, silent type to relax.DNA Repair (Amst). 2010 Dec 10;9(12):1273-82. doi: 10.1016/j.dnarep.2010.09.013. Epub 2010 Oct 30. DNA Repair (Amst). 2010. PMID: 21036673 Review.
-
The heterochromatin protein 1 (HP1) family: put away a bias toward HP1.Mol Cells. 2008 Sep 30;26(3):217-27. Epub 2008 Jul 30. Mol Cells. 2008. PMID: 18664736 Review.
Cited by
-
Functional Analysis Identifies Damaging CHEK2 Missense Variants Associated with Increased Cancer Risk.Cancer Res. 2022 Feb 15;82(4):615-631. doi: 10.1158/0008-5472.CAN-21-1845. Cancer Res. 2022. PMID: 34903604 Free PMC article.
-
Genome-wide redistribution of H3K27me3 is linked to genotoxic stress and defective growth.Proc Natl Acad Sci U S A. 2015 Nov 17;112(46):E6339-48. doi: 10.1073/pnas.1511377112. Epub 2015 Nov 2. Proc Natl Acad Sci U S A. 2015. PMID: 26578794 Free PMC article.
-
Non-Coding RNA: Sequence-Specific Guide for Chromatin Modification and DNA Damage Signaling.Front Genet. 2015 Nov 13;6:320. doi: 10.3389/fgene.2015.00320. eCollection 2015. Front Genet. 2015. PMID: 26617633 Free PMC article. Review.
-
Interplay and Dynamics of Chromatin Architecture and DNA Damage Response: An Overview.Cancers (Basel). 2025 Mar 11;17(6):949. doi: 10.3390/cancers17060949. Cancers (Basel). 2025. PMID: 40149285 Free PMC article. Review.
-
Chromatin modifications and the DNA damage response to ionizing radiation.Front Oncol. 2013 Jan 22;2:214. doi: 10.3389/fonc.2012.00214. eCollection 2012. Front Oncol. 2013. PMID: 23346550 Free PMC article.
References
-
- Friedman J. R., Fredericks W. J., Jensen D. E., Speicher D. W., Huang X. P., Neilson E. G., Rauscher F. J., 3rd (1996) KAP-1, a novel co-repressor for the highly conserved KRAB repression domain. Genes Dev. 10, 2067–2078 - PubMed
-
- Ryan R. F., Schultz D. C., Ayyanathan K., Singh P. B., Friedman J. R., Fredericks W. J., Rauscher F. J., 3rd (1999) KAP-1 co-repressor protein interacts and colocalizes with heterochromatic and euchromatic HP1 proteins: a potential role for Krüppel-associated box-zinc finger proteins in heterochromatin-mediated gene silencing. Mol. Cell Biol. 19, 4366–4378 - PMC - PubMed
-
- Goodarzi A. A., Jeggo P., Lobrich M. (2010) The influence of heterochromatin on DNA double-strand break repair: getting the strong, silent type to relax. DNA Repair 9, 1273–1282 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

