Stereoselective preparation of cyclobutanes with four different substituents: total synthesis and structural revision of pipercyclobutanamide A and piperchabamide G
- PMID: 22715150
- PMCID: PMC3444511
- DOI: 10.1002/anie.201203379
Stereoselective preparation of cyclobutanes with four different substituents: total synthesis and structural revision of pipercyclobutanamide A and piperchabamide G
Abstract
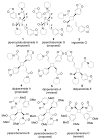
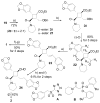
A general strategy was developed for the diastereo- and enantioselective synthesis of cyclobutanes with four different substituents. It consists of three transition metal-catalyzed reactions — a RhII-catalyzed cyclopropanation, a AgI-catalyzed regioselective and stereospecific ring expansion, and a RhI-catalyzed addition reaction. Structures of pipercyclobutanamide A and piperchabamide G were synthesized and revised.
Figures
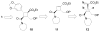

Similar articles
-
Sequential C(sp3)-H arylation and olefination: total synthesis of the proposed structure of pipercyclobutanamide A.Angew Chem Int Ed Engl. 2012 Jul 23;51(30):7507-10. doi: 10.1002/anie.201203897. Epub 2012 Jun 19. Angew Chem Int Ed Engl. 2012. PMID: 22715157 Free PMC article.
-
Synthesis of chiral cyclobutanes via rhodium/diene-catalyzed asymmetric 1,4-addition: a dramatic ligand effect on the diastereoselectivity.Chem Commun (Camb). 2015 May 25;51(42):8773-6. doi: 10.1039/c5cc02023a. Chem Commun (Camb). 2015. PMID: 25912603
-
Column chromatography-free solution-phase synthesis of a natural piper-amide-like compound library.ACS Comb Sci. 2013 Apr 8;15(4):208-15. doi: 10.1021/co400003d. Epub 2013 Mar 4. ACS Comb Sci. 2013. PMID: 23458110
-
Rhodium(II)-catalysed intramolecular C-H insertion α- to oxygen: reactivity, selectivity and applications to natural product synthesis.Org Biomol Chem. 2015 Jun 21;13(23):6419-31. doi: 10.1039/c5ob00311c. Epub 2015 May 12. Org Biomol Chem. 2015. PMID: 25965780 Review.
-
Guiding principles for site selective and stereoselective intermolecular C-H functionalization by donor/acceptor rhodium carbenes.Chem Soc Rev. 2011 Apr;40(4):1857-69. doi: 10.1039/c0cs00217h. Epub 2011 Mar 1. Chem Soc Rev. 2011. PMID: 21359404 Review.
Cited by
-
Natural Products Modulating Angiotensin Converting Enzyme 2 (ACE2) as Potential COVID-19 Therapies.Front Pharmacol. 2021 May 3;12:629935. doi: 10.3389/fphar.2021.629935. eCollection 2021. Front Pharmacol. 2021. PMID: 34012391 Free PMC article. Review.
-
Generation of rhodium(I) carbenes from ynamides and their reactions with alkynes and alkenes.J Am Chem Soc. 2013 Jun 5;135(22):8201-4. doi: 10.1021/ja4047069. Epub 2013 May 23. J Am Chem Soc. 2013. PMID: 23701315 Free PMC article.
-
Intramolecular thermal stepwise [2 + 2] cycloadditions: investigation of a stereoselective synthesis of [n.2.0]-bicyclolactones.Org Biomol Chem. 2016 Oct 12;14(40):9554-9559. doi: 10.1039/c6ob01661h. Org Biomol Chem. 2016. PMID: 27722445 Free PMC article.
-
Enantioselective Crossed Photocycloadditions of Styrenic Olefins by Lewis Acid Catalyzed Triplet Sensitization.Angew Chem Int Ed Engl. 2017 Sep 18;56(39):11891-11895. doi: 10.1002/anie.201706975. Epub 2017 Aug 30. Angew Chem Int Ed Engl. 2017. PMID: 28776908 Free PMC article.
-
A Mixed-Ligand Chiral Rhodium(II) Catalyst Enables the Enantioselective Total Synthesis of Piperarborenine B.Angew Chem Int Ed Engl. 2016 Apr 11;55(16):4983-7. doi: 10.1002/anie.201600766. Epub 2016 Mar 16. Angew Chem Int Ed Engl. 2016. PMID: 26991451 Free PMC article.
References
-
-
For selected reviews on four-membered ring containing natural products, see: Hansen TV, Stenstrom Y. Organic Synthesis: Theory and Applications. Vol. 5. Elsevier Science Ltd; 2001. Naturally Occurring Cyclobutanes; p. 1.Dembitsky VM. J Nat Med. 2008;62:1.
-
-
- Fujiwara Y, Naithou K, Miyazaki T, Hashimoto K, Mori K, Yamamoto Y. Tetrahedron Lett. 2001;42:2497.
-
- Tsukamoto S, Cha BC, Ohta T. Tetrahedron. 2002;58:1667.
-
- Tsukamoto S, Tomise K, Miyakawa K, Cha BC, Abe T, Hamada T, Hirota H, Ohta T. Bioorg Med Chem. 2002;10:2981. - PubMed
-
- Wei K, Li W, Koike K, Chen YJ, Nikaido T. J Org Chem. 2005;70:1164. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

