Behavior-related pauses in simple-spike activity of mouse Purkinje cells are linked to spike rate modulation
- PMID: 22723707
- PMCID: PMC3403286
- DOI: 10.1523/JNEUROSCI.4969-11.2012
Behavior-related pauses in simple-spike activity of mouse Purkinje cells are linked to spike rate modulation
Abstract
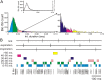
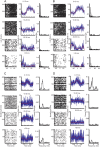
Purkinje cells (PCs) in the mammalian cerebellum express high-frequency spontaneous activity with average spike rates between 30 and 200 Hz. Cerebellar nuclear (CN) neurons receive converging input from many PCs, resulting in a continuous barrage of inhibitory inputs. It has been hypothesized that pauses in PC activity trigger increases in CN spiking activity. A prediction derived from this hypothesis is that pauses in PC simple-spike activity represent relevant behavioral or sensory events. Here, we asked whether pauses in the simple-spike activity of PCs related to either fluid licking or respiration, play a special role in representing information about behavior. Both behaviors are widely represented in cerebellar PC simple-spike activity. We recorded PC activity in the vermis and lobus simplex of head-fixed mice while monitoring licking and respiratory behavior. Using cross-correlation and Granger causality analysis, we examined whether short interspike intervals (ISIs) had a different temporal relationship to behavior than long ISIs or pauses. Behavior-related simple-spike pauses occurred during low-rate simple-spike activity in both licking- and breathing-related PCs. Granger causality analysis revealed causal relationships between simple-spike pauses and behavior. However, the same results were obtained from an analysis of surrogate spike trains with gamma ISI distributions constructed to match rate modulations of behavior-related Purkinje cells. Our results therefore suggest that the occurrence of pauses in simple-spike activity does not represent additional information about behavioral or sensory events that goes beyond the simple-spike rate modulations.
Figures




References
-
- Aizenman CD, Linden DJ. Regulation of the rebound depolarization and spontaneous firing patterns of deep nuclear neurons in slices of rat cerebellum. J Neurophysiol. 1999;82:1697–1709. - PubMed
-
- Antkowiak B, Heck DH. Effects of the volatile anaesthetic enflurane on spontaneous discharge rate and GABA A-mediated inhibition of Purkinje cells in rat cerebellar slices. J Neurophysiol. 1997;77:2525–2538. - PubMed
-
- De Schutter E, Steuber V. Patterns and pauses in Purkinje cell simple spike trains: experiments, modeling and theory. Neuroscience. 2009;162:816–826. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
