Sustained therapeutic reversal of Huntington's disease by transient repression of huntingtin synthesis
- PMID: 22726834
- PMCID: PMC3383626
- DOI: 10.1016/j.neuron.2012.05.009
Sustained therapeutic reversal of Huntington's disease by transient repression of huntingtin synthesis
Abstract
The primary cause of Huntington's disease (HD) is expression of huntingtin with a polyglutamine expansion. Despite an absence of consensus on the mechanism(s) of toxicity, diminishing the synthesis of mutant huntingtin will abate toxicity if delivered to the key affected cells. With antisense oligonucleotides (ASOs) that catalyze RNase H-mediated degradation of huntingtin mRNA, we demonstrate that transient infusion into the cerebrospinal fluid of symptomatic HD mouse models not only delays disease progression but mediates a sustained reversal of disease phenotype that persists longer than the huntingtin knockdown. Reduction of wild-type huntingtin, along with mutant huntingtin, produces the same sustained disease reversal. Similar ASO infusion into nonhuman primates is shown to effectively lower huntingtin in many brain regions targeted by HD pathology. Rather than requiring continuous treatment, our findings establish a therapeutic strategy for sustained HD disease reversal produced by transient ASO-mediated diminution of huntingtin synthesis.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures




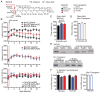
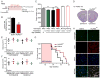

Comment in
-
"Huntingtin holiday": progress toward an antisense therapy for Huntington's disease.Neuron. 2012 Jun 21;74(6):964-6. doi: 10.1016/j.neuron.2012.06.001. Neuron. 2012. PMID: 22726826 Free PMC article.
-
Neurodegenerative disorders: Reversing Huntington's disease.Nat Rev Drug Discov. 2012 Aug;11(8):600. doi: 10.1038/nrd3804. Nat Rev Drug Discov. 2012. PMID: 22850779 No abstract available.
-
Making sense out of antisense in Huntington's disease.CNS Neurol Disord Drug Targets. 2012 Sep;11(6):647-8. doi: 10.2174/187152712803581038. CNS Neurol Disord Drug Targets. 2012. PMID: 22998135 No abstract available.
References
-
- Bennett CF, Swayze EE. RNA targeting therapeutics: molecular mechanisms of antisense oligonucleotides as a therapeutic platform. Annu Rev Pharmacol Toxicol. 2010;50:259–293. - PubMed
-
- Boillee S, Yamanaka K, Lobsiger CS, Copeland NG, Jenkins NA, Kassiotis G, Kollias G, Cleveland DW. Onset and progression in inherited ALS determined by motor neurons and microglia. Science. 2006;312:1389–1392. - PubMed
-
- Brooks E, Arrasate M, Cheung K, Finkbeiner SM. Using antibodies to analyze polyglutamine stretches. Methods Mol Biol. 2004;277:103–128. - PubMed
-
- Carroll JB, Warby SC, Southwell AL, Doty CN, Greenlee S, Skotte N, Hung G, Bennett CF, Freier SM, Hayden MR. Potent and selective antisense oligonucleotides targeting single-nucleotide polymorphisms in the huntington disease gene/allele-specific silencing of mutant huntingtin. Mol Ther. 2011;19:2178–2185. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

