Odor representations in olfactory cortex: distributed rate coding and decorrelated population activity
- PMID: 22726838
- PMCID: PMC3383608
- DOI: 10.1016/j.neuron.2012.04.021
Odor representations in olfactory cortex: distributed rate coding and decorrelated population activity
Abstract
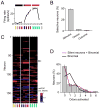
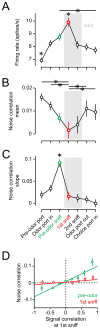
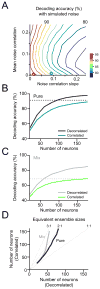
How information encoded in neuronal spike trains is used to guide sensory decisions is a fundamental question. In olfaction, a single sniff is sufficient for fine odor discrimination but the neural representations on which olfactory decisions are based are unclear. Here, we recorded neural ensemble activity in the anterior piriform cortex (aPC) of rats performing an odor mixture categorization task. We show that odors evoke transient bursts locked to sniff onset and that odor identity can be better decoded using burst spike counts than by spike latencies or temporal patterns. Surprisingly, aPC ensembles also exhibited near-zero noise correlations during odor stimulation. Consequently, fewer than 100 aPC neurons provided sufficient information to account for behavioral speed and accuracy, suggesting that behavioral performance limits arise downstream of aPC. These findings demonstrate profound transformations in the dynamics of odor representations from the olfactory bulb to cortex and reveal likely substrates for odor-guided decisions.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures


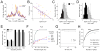


References
-
- Averbeck BB, Latham PE, Pouget A. Neural correlations, population coding and computation. Nat Rev Neurosci. 2006;7:358–366. - PubMed
-
- Blumhagen F, Zhu P, Shum J, Scharer YP, Yaksi E, Deisseroth K, Friedrich RW. Neuronal filtering of multiplexed odour representations. Nature. 2011;479:493–498. - PubMed
-
- Britten KH, Newsome WT, Shadlen MN, Celebrini S, Movshon JA. A relationship between behavioral choice and the visual responses of neurons in macaque MT. Vis Neurosci. 1996;13:87–100. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

