Doublecortin recognizes the 13-protofilament microtubule cooperatively and tracks microtubule ends
- PMID: 22727374
- PMCID: PMC3951992
- DOI: 10.1016/j.devcel.2012.05.006
Doublecortin recognizes the 13-protofilament microtubule cooperatively and tracks microtubule ends
Abstract
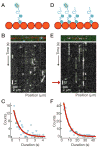
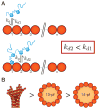
Neurons, like all cells, face the problem that tubulin forms microtubules with too many or too few protofilaments (pfs). Cells overcome this heterogeneity with the γ-tubulin ring complex, which provides a nucleation template for 13-pf microtubules. Doublecortin (DCX), a protein that stabilizes microtubules in developing neurons, also nucleates 13-pf microtubules in vitro. Using fluorescence microscopy assays, we show that the binding of DCX to microtubules is optimized for the lateral curvature of the 13-pf lattice. This sensitivity depends on a cooperative interaction wherein DCX molecules decrease the dissociation rate of their neighbors. Mutations in DCX found in patients with subcortical band heterotopia weaken these cooperative interactions. Using assays with dynamic microtubules, we discovered that DCX binds to polymerization intermediates at growing microtubule ends. These results support a mechanism for stabilizing 13-pf microtubules that allows DCX to template new 13-pf microtubules through associations with the sides of the microtubule lattice.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures

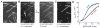


Comment in
-
Measuring microtubule thickness: an exercise in cooperativity.Dev Cell. 2012 Jul 17;23(1):1-2. doi: 10.1016/j.devcel.2012.06.010. Dev Cell. 2012. PMID: 22814594
References
-
- Ashford A, Andersen S, Hyman AA. Preparation of Tubulin from Bovine Brain. In: Celis J, editor. Cell Biology, a Laboratory Handbook. Vol. 2. Academic Press; New York: 1998. pp. 205–212.
-
- Bai J, Ramos RL, Ackman JB, Thomas AM, Lee RV, LoTurco JJ. RNAi reveals doublecortin is required for radial migration in rat neocortex. Nat Neurosci. 2003;6(12):1277–83. - PubMed
-
- Bieling P, Laan L, Schek H, Munteanu EL, Sandblad L, Dogterom M, Brunner D, Surrey T. Reconstitution of a microtubule plus-end tracking system in vitro. Nature. 2007;450(7172):1100–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

