DNA circuits as amplifiers for the detection of nucleic acids on a paperfluidic platform
- PMID: 22729075
- PMCID: PMC3454488
- DOI: 10.1039/c2lc40373k
DNA circuits as amplifiers for the detection of nucleic acids on a paperfluidic platform
Abstract
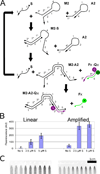
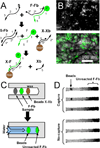
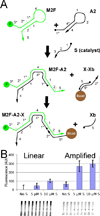
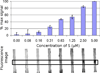
This article describes the use of non-enzymatic nucleic acid circuits based on strand exchange reactions to detect target sequences on a paperfluidic platform. The DNA circuits that were implemented include a non-enzymatic amplifier and transduction to a fluorescent reporter; these yield an order of magnitude improvement in detection of an input nucleic acid signal. To further improve signal amplification and detection, we integrated the enzyme-free amplifier with loop-mediated isothermal amplification (LAMP). By bridging the gap between the low concentrations of LAMP amplicons and the limits of fluorescence detection, the non-enzymatic amplifier allowed us to detect as few as 1200 input templates in a paperfluidic format.
Figures


References
-
- Carrilho E, Martinez AW, Whitesides GM. Anal. Chem. 2009;81:7091–7095. - PubMed
-
- Su S, Ali MM, Filipe CDM, Li Y, Pelton R. Biomacromolecules. 2008;9:935–941. - PubMed
-
- Martinez AW, Phillips ST, Nie Z, Cheng C-M, Carrilho E, Wiley BJ, Whitesides GM. Lab Chip. 2010;10:3428.
-
- Fenton EM, Mascarenas MR, LoÌ pez GP, Sibbett SS. ACS Appl. Mater. Interfaces. 2009;1:124–129. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

