ATP induces the death of developing avian retinal neurons in culture via activation of P2X7 and glutamate receptors
- PMID: 22733428
- PMCID: PMC3568422
- DOI: 10.1007/s11302-012-9324-5
ATP induces the death of developing avian retinal neurons in culture via activation of P2X7 and glutamate receptors
Abstract
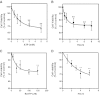
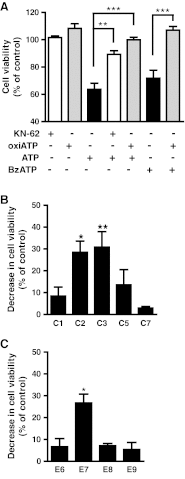
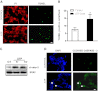
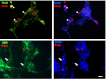
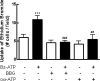
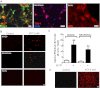
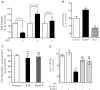
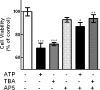
Previous data suggest that nucleotides are important mitogens in the developing retina. Here, the effect of ATP on the death of cultured chick embryo retina cells was investigated. In cultures obtained from retinas of 7-day-old chick embryos (E7) that were cultivated for 2 days (E7C2), both ATP and BzATP induced a ∼30 % decrease in cell viability that was time- and dose-dependent and that could be blocked by 0.2 mM oxidized ATP or 0.3 μM KN-62. An increase in cleaved caspase-3 levels and in the number of TUNEL-positive cells was observed when cultures were incubated with 3 mM ATP and immunolabeling for cleaved-caspase 3 was observed over neurons but not over glial cells. ATP-dependent cell death was developmentally regulated, the maximal levels being detected by E7C2-3. Nucleotides were able to increase neuronal ethidium bromide and sulforhodamine B uptake in mixed and purified neuronal cultures, an effect that was blocked by the antagonists Brilliant Blue G and oxidized ATP. In contrast, nucleotide-induced cell death was observed only in mixed cultures, but not in purified cultures of neurons or glia. ATP-induced neuronal death was blocked by the glutamatergic antagonists MK801 and DNQX and activation of P2X7 receptors by ATP decreased the uptake of [(3)H]-D-aspartate by cultured glial cells with a concomitant accumulation of it in the extracellular medium. These results suggest that ATP induces apoptosis of chick embryo retinal neurons in culture through activation of P2X7 and glutamate ionotropic receptors. Involvement of a P2X7 receptor-mediated inhibition of the glial uptake of glutamate is suggested.
Figures


References
-
- Frade JM, Bovolenta P, Martínez-Morales JR, Arribas A, Barbas JA, Rodríguez-Tébar A. Control of early cell death by BDNF in the chick retina. Development. 1997;124:3313–3320. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

