Investigating ligand-receptor interactions at bilayer surface using electronic absorption spectroscopy and fluorescence resonance energy transfer
- PMID: 22734511
- PMCID: PMC3439585
- DOI: 10.1021/la300724z
Investigating ligand-receptor interactions at bilayer surface using electronic absorption spectroscopy and fluorescence resonance energy transfer
Abstract
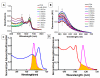
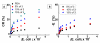
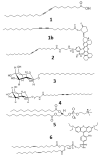
We investigate interactions between receptors and ligands at bilayer surface of polydiacetylene (PDA) liposomal nanoparticles using changes in electronic absorption spectroscopy and fluorescence resonance energy transfer (FRET). We study the effect of mode of linkage (covalent versus noncovalent) between the receptor and liposome bilayer. We also examine the effect of size-dependent interactions between liposome and analyte through electronic absorption and FRET responses. Glucose (receptor) molecules were either covalently or noncovalently attached at the bilayer of nanoparticles, and they provided selectivity for molecular interactions between glucose and glycoprotein ligands of E. coli. These interactions induced stress on conjugated PDA chain which resulted in changes (blue to red) in the absorption spectrum of PDA. The changes in electronic absorbance also led to changes in FRET efficiency between conjugated PDA chains (acceptor) and fluorophores (Sulphorhodamine-101) (donor) attached to the bilayer surface. Interestingly, we did not find significant differences in UV-vis and FRET responses for covalently and noncovalently bound glucose to liposomes following their interactions with E. coli. We attributed these results to close proximity of glucose receptor molecules to the liposome bilayer surface such that induced stress were similar in both the cases. We also found that PDA emission from direct excitation mechanism was ~2-10 times larger than that of the FRET-based response. These differences in emission signals were attributed to three major reasons: nonspecific interactions between E. coli and liposomes, size differences between analyte and liposomes, and a much higher PDA concentration with respect to sulforhodamine (SR-101). We have proposed a model to explain our experimental observations. Our fundamental studies reported here will help in enhancing our knowledge regarding interactions involved between soft particles at molecular levels.
Figures


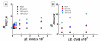
Similar articles
-
Real-time monitoring of ligand-receptor interactions with fluorescence resonance energy transfer.J Vis Exp. 2012 Aug 20;(66):e3805. doi: 10.3791/3805. J Vis Exp. 2012. PMID: 22929922 Free PMC article.
-
Fluorescence resonance energy transfer in polydiacetylene liposomes.J Phys Chem B. 2008 Oct 23;112(42):13263-72. doi: 10.1021/jp804640p. Epub 2008 Sep 25. J Phys Chem B. 2008. PMID: 18816092 Free PMC article.
-
Enhancing the emission of polydiacetylene sensing materials through fluorophore addition and energy transfer.J Fluoresc. 2008 Mar;18(2):461-71. doi: 10.1007/s10895-007-0287-9. Epub 2007 Dec 20. J Fluoresc. 2008. PMID: 18097739
-
Structures and strategies for enhanced sensitivity of polydiacetylene(PDA) based biosensor platforms.Biosens Bioelectron. 2021 Jun 1;181:113120. doi: 10.1016/j.bios.2021.113120. Epub 2021 Mar 2. Biosens Bioelectron. 2021. PMID: 33714858 Review.
-
Biomolecule-Functionalized Smart Polydiacetylene for Biomedical and Environmental Sensing.Molecules. 2018 Jan 4;23(1):107. doi: 10.3390/molecules23010107. Molecules. 2018. PMID: 29300355 Free PMC article. Review.
Cited by
-
Responsive Polydiacetylene Vesicles for Biosensing Microorganisms.Sensors (Basel). 2018 Feb 15;18(2):599. doi: 10.3390/s18020599. Sensors (Basel). 2018. PMID: 29462870 Free PMC article. Review.
-
Biosensors for the Detection of Food Pathogens.Foods. 2014 Sep 2;3(3):511-526. doi: 10.3390/foods3030511. Foods. 2014. PMID: 28234334 Free PMC article. Review.
-
Micro-motors: A motile bacteria based system for liposome cargo transport.Sci Rep. 2016 Jul 5;6:29369. doi: 10.1038/srep29369. Sci Rep. 2016. PMID: 27377152 Free PMC article.
-
COVID-19 and Kidney Disease: Molecular Determinants and Clinical Implications in Renal Cancer.Eur Urol Focus. 2020 Sep 15;6(5):1086-1096. doi: 10.1016/j.euf.2020.06.002. Epub 2020 Jun 9. Eur Urol Focus. 2020. PMID: 32540268 Free PMC article. Review.
-
Second-Order Scattering Quenching in Fluorescence Spectra of Natural Humates as a Tracer of Formation Stable Supramolecular System for the Delivery of Poorly Soluble Antiviral Drugs on the Example of Mangiferin and Favipiravir.Pharmaceutics. 2022 Mar 31;14(4):767. doi: 10.3390/pharmaceutics14040767. Pharmaceutics. 2022. PMID: 35456601 Free PMC article.
References
-
- Karlsson KA. Microbial recognition of target-cell glycoconjugates Curr. Opin. Struct. Biol. 1995;5:622. - PubMed
-
- Guo CX, Boullanger P, Liu T, Jiang L. Size effect of polydiacetylene vesicles functionalized with glycolipids on their colorimetric detection ability. J. Phys. Chem. B. 2005;109:18765–18771. - PubMed
-
- Sharon N, Lis H. Lectins as cell recognition molecules. Science. 1989;246:227. - PubMed
-
- Spevak W, Nagy JO, Charych DH. Molecular assemblies of functionalized polydiacetylenes. Adv. Mater. 1995;7:85–89.
-
- Kim TH, Swager TM. A fluorescent self-amplifying wavelength-responsive sensory polymer for fluoride ions. Angew. Chem. Int. Ed. 2003;42:4803. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

