Sufficient virus-neutralizing antibody in the central nerve system improves the survival of rabid rats
- PMID: 22734518
- PMCID: PMC3431253
- DOI: 10.1186/1423-0127-19-61
Sufficient virus-neutralizing antibody in the central nerve system improves the survival of rabid rats
Abstract
Background: Rabies is known to be lethal in human. Treatment with passive immunity for the rabies is effective only when the patients have not shown the central nerve system (CNS) signs. The blood-brain barrier (BBB) is a complex functional barrier that may compromise the therapeutic development in neurological diseases. The goal of this study is to determine the change of BBB integrity and to assess the therapeutic possibility of enhancing BBB permeability combined with passive immunity in the late stage of rabies virus infection.
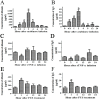
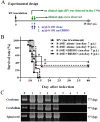
Methods: The integrity of BBB permeability in rats was measured by quantitative ELISA for total IgG and albumin levels in the cerebrospinal fluid (CSF) and by exogenously applying Evans blue as a tracer. Western blotting of occludin and ZO-1, two tight junction proteins, was used to assess the molecular change of BBB structure.The breakdown of BBB with hypertonic arabinose, recombinant tumor necrosis factor-alpha (rTNF-γ), and focused ultrasound (FUS) were used to compare the extent of BBB disruption with rabies virus infection. Specific humoral immunity was analyzed by immunofluorescent assay and rapid fluorescent focus inhibition test. Virus-neutralizing monoclonal antibody (mAb) 8-10E was administered to rats with hypertonic breakdown of BBB as a passive immunotherapy to prevent the death from rabies.
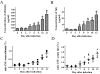
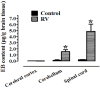
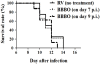
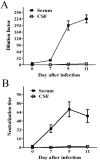
Results: The BBB permeability was altered on day 7 post-infection. Increased BBB permeability induced by rabies virus infection was observed primarily in the cerebellum and spinal cord. Occludin was significantly decreased in both the cerebral cortex and cerebellum. The rabies virus-specific antibody was not strongly elicited even in the presence of clinical signs. Disruption of BBB had no direct association with the lethal outcome of rabies. Passive immunotherapy with virus-neutralizing mAb 8-10E with the hypertonic breakdown of BBB prolonged the survival of rabies virus-infected rats.
Conclusions: We demonstrated that the BBB permeability was altered in a rat model with rabies virus inoculation. Delivery of neutralizing mAb to the infected site in brain combined with effective breakdown of BBB could be an aggressive but feasible therapeutic mode in rabies when the CNS infection has been established.
Figures

Similar articles
-
Enhancement of blood-brain barrier permeability and reduction of tight junction protein expression are modulated by chemokines/cytokines induced by rabies virus infection.J Virol. 2014 May;88(9):4698-710. doi: 10.1128/JVI.03149-13. Epub 2014 Feb 12. J Virol. 2014. PMID: 24522913 Free PMC article.
-
Regional differences in blood-brain barrier permeability changes and inflammation in the apathogenic clearance of virus from the central nervous system.J Immunol. 2006 Jun 15;176(12):7666-75. doi: 10.4049/jimmunol.176.12.7666. J Immunol. 2006. PMID: 16751414
-
Enhancement of blood-brain barrier permeability is required for intravenously administered virus neutralizing antibodies to clear an established rabies virus infection from the brain and prevent the development of rabies in mice.Antiviral Res. 2014 Oct;110:132-41. doi: 10.1016/j.antiviral.2014.07.013. Epub 2014 Aug 7. Antiviral Res. 2014. PMID: 25108172 Free PMC article.
-
Innate Immune Signaling and Role of Glial Cells in Herpes Simplex Virus- and Rabies Virus-Induced Encephalitis.Viruses. 2021 Nov 25;13(12):2364. doi: 10.3390/v13122364. Viruses. 2021. PMID: 34960633 Free PMC article. Review.
-
Role of chemokines in rabies pathogenesis and protection.Adv Virus Res. 2011;79:73-89. doi: 10.1016/B978-0-12-387040-7.00005-6. Adv Virus Res. 2011. PMID: 21601043 Free PMC article. Review.
Cited by
-
Characterisation of a Live-Attenuated Rabies Virus Expressing a Secreted scFv for the Treatment of Rabies.Viruses. 2023 Jul 31;15(8):1674. doi: 10.3390/v15081674. Viruses. 2023. PMID: 37632016 Free PMC article.
-
Generation of recombinant rabies Virus CVS-11 expressing eGFP applied to the rapid virus neutralization test.Viruses. 2014 Apr 4;6(4):1578-89. doi: 10.3390/v6041578. Viruses. 2014. PMID: 24714411 Free PMC article.
-
Rabies Control and Treatment: From Prophylaxis to Strategies with Curative Potential.Viruses. 2016 Oct 28;8(11):279. doi: 10.3390/v8110279. Viruses. 2016. PMID: 27801824 Free PMC article. Review.
-
Enhancement of blood-brain barrier permeability and reduction of tight junction protein expression are modulated by chemokines/cytokines induced by rabies virus infection.J Virol. 2014 May;88(9):4698-710. doi: 10.1128/JVI.03149-13. Epub 2014 Feb 12. J Virol. 2014. PMID: 24522913 Free PMC article.
-
Immunization with recombinant rabies virus expressing Interleukin-18 exhibits enhanced immunogenicity and protection in mice.Oncotarget. 2017 Sep 16;8(53):91505-91515. doi: 10.18632/oncotarget.21065. eCollection 2017 Oct 31. Oncotarget. 2017. PMID: 29207661 Free PMC article.
References
-
- WHO. Expert Consultation on rabies. World Health Organ Tech Rep Ser. 2005;931:1–88. back cover. - PubMed
-
- Jackson AC. Therapy of rabies encephalitis. Biomedica. 2009;29:169–176. - PubMed
-
- Schmiedel S, Panning M, Lohse A, Kreymann KG, Gerloff C, Burchard G, Drosten C, Schmiedel S, Panning M, Lohse A, Kreymann KG, Gerloff C, Burchard G, Drosten C. Case report on fatal human rabies infection in Hamburg, Germany, March 2007. Euro Surveill. 2007;12:E070531–E070535. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

