Rapid uptake and degradation of CXCL12 depend on CXCR7 carboxyl-terminal serine/threonine residues
- PMID: 22736769
- PMCID: PMC3436560
- DOI: 10.1074/jbc.M111.335679
Rapid uptake and degradation of CXCL12 depend on CXCR7 carboxyl-terminal serine/threonine residues
Abstract
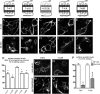
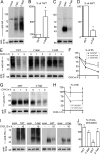
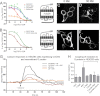
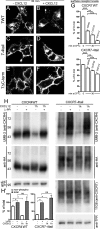
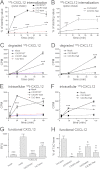
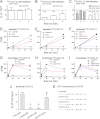
CXCL12 signaling through G protein-coupled CXCR4 regulates cell migration during ontogenesis and disease states including cancer and inflammation. The second CXCL12-receptor CXCR7 modulates the CXCL12/CXCR4 pathway by acting as a CXCL12 scavenger and exerts G protein-independent functions. Given the distinct properties of CXCR4 and CXCR7, we hypothesized that the distinct C-terminal domains differently regulate receptor trafficking and stability. Here, we examined epitope-tagged wild type and C-terminal mutant receptors in human embryonic kidney cells (HEK293) with respect to trafficking, stability, (125)I-CXCL12 degradation, and G protein-coupling. The 24 CXCR7 C-terminal residues were sufficient to promote rapid spontaneous internalization. Replacement of the CXCR7 C terminus with that of CXCR4 (CXCR7-4tail mutant) abolished spontaneous internalization but permitted ligand-induced internalization and phosphorylation at the heterologous domain. The reverse tail-swap caused ligand-independent internalization of the resulting CXCR4-7tail mutant. Receptor-mediated (125)I-CXCL12 uptake and release of (125)I-CXCL12 degradation products were accelerated with receptors bearing the CXCR7 C terminus and impaired after conversion of CXCR7 C-terminal serine/threonine residues into alanines. C-terminal lysine residues were dispensable for plasma membrane targeting and the CXCL12 scavenger function but involved in constitutive degradation of CXCR7. Although the CXCR7 C terminus abolished G protein coupling in the CXCR4-7tail mutant, replacement of the CXCR7 C terminus, CXCR7 second intracellular loop, or both domains with the corresponding CXCR4 domain did not result in a G protein-coupled CXCR7 chimera. Taken together, we provide evidence that the CXCR7 C terminus influences the ligand-uptake/degradation rate, G protein coupling, and receptor stability. Regulatory pathways targeting CXCR7 C-terminal serine/threonine sites may control the CXCL12 scavenger activity of CXCR7.
Figures

Similar articles
-
The peptidomimetic CXCR4 antagonist TC14012 recruits beta-arrestin to CXCR7: roles of receptor domains.J Biol Chem. 2010 Dec 3;285(49):37939-43. doi: 10.1074/jbc.C110.147470. Epub 2010 Oct 18. J Biol Chem. 2010. PMID: 20956518 Free PMC article.
-
CXCR4 and CXCR7 transduce through mTOR in human renal cancer cells.Cell Death Dis. 2014 Jul 3;5(7):e1310. doi: 10.1038/cddis.2014.269. Cell Death Dis. 2014. PMID: 24991762 Free PMC article.
-
CXCR7/CXCR4 heterodimer constitutively recruits beta-arrestin to enhance cell migration.J Biol Chem. 2011 Sep 16;286(37):32188-97. doi: 10.1074/jbc.M111.277038. Epub 2011 Jul 5. J Biol Chem. 2011. PMID: 21730065 Free PMC article.
-
CXCL12-CXCR4/CXCR7 Axis in Colorectal Cancer: Therapeutic Target in Preclinical and Clinical Studies.Int J Mol Sci. 2021 Jul 9;22(14):7371. doi: 10.3390/ijms22147371. Int J Mol Sci. 2021. PMID: 34298991 Free PMC article. Review.
-
Drug Design Targeting the CXCR4/CXCR7/CXCL12 Pathway.Curr Top Med Chem. 2016;16(13):1441-51. doi: 10.2174/1568026615666150915120218. Curr Top Med Chem. 2016. PMID: 26369824 Review.
Cited by
-
The atypical chemokine receptor 3 interacts with Connexin 43 inhibiting astrocytic gap junctional intercellular communication.Nat Commun. 2020 Sep 25;11(1):4855. doi: 10.1038/s41467-020-18634-y. Nat Commun. 2020. PMID: 32978390 Free PMC article.
-
Osteotropism of neuroendocrine tumors: role of the CXCL12/ CXCR4 pathway in promoting EMT in vitro.Oncotarget. 2017 Apr 4;8(14):22534-22549. doi: 10.18632/oncotarget.15122. Oncotarget. 2017. PMID: 28186979 Free PMC article.
-
Stromal derived factor 1α: a chemokine that delivers a two-pronged defence of the myocardium.Pharmacol Ther. 2014 Sep;143(3):305-15. doi: 10.1016/j.pharmthera.2014.03.009. Epub 2014 Apr 1. Pharmacol Ther. 2014. PMID: 24704323 Free PMC article. Review.
-
The US27 gene product of human cytomegalovirus enhances signaling of host chemokine receptor CXCR4.Virology. 2013 May 10;439(2):122-31. doi: 10.1016/j.virol.2013.02.006. Epub 2013 Mar 13. Virology. 2013. PMID: 23490053 Free PMC article.
-
Mutational Analysis of Atypical Chemokine Receptor 3 (ACKR3/CXCR7) Interaction with Its Chemokine Ligands CXCL11 and CXCL12.J Biol Chem. 2017 Jan 6;292(1):31-42. doi: 10.1074/jbc.M116.762252. Epub 2016 Nov 14. J Biol Chem. 2017. PMID: 27875312 Free PMC article.
References
-
- Sierro F., Biben C., Martínez-Muñoz L., Mellado M., Ransohoff R. M., Li M., Woehl B., Leung H., Groom J., Batten M., Harvey R. P., Martínez-A C., Mackay C. R., Mackay F. (2007) Disrupted cardiac development but normal hematopoiesis in mice deficient in the second CXCL12/SDF-1 receptor, CXCR7. Proc. Natl. Acad. Sci. U.S.A. 104, 14759–14764 - PMC - PubMed
-
- Zou Y. R., Kottmann A. H., Kuroda M., Taniuchi I., Littman D. R. (1998) Function of the chemokine receptor CXCR4 in hematopoiesis and in cerebellar development. Nature 393, 595–599 - PubMed
-
- Nagasawa T., Hirota S., Tachibana K., Takakura N., Nishikawa S., Kitamura Y., Yoshida N., Kikutani H., Kishimoto T. (1996) Defects of B-cell lymphopoiesis and bone-marrow myelopoiesis in mice lacking the CXC chemokine PBSF/SDF-1. Nature 382, 635–638 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

