Enhanced anti-HIV efficacy of indinavir after inclusion in CD4-targeted lipid nanoparticles
- PMID: 22743598
- PMCID: PMC3551348
- DOI: 10.1097/QAI.0b013e3182653c1f
Enhanced anti-HIV efficacy of indinavir after inclusion in CD4-targeted lipid nanoparticles
Abstract
Background: Combination drug therapy has reduced plasma HIV to undetectable levels; however, drug-sensitive virus persists in patients' lymphoid tissue. We have reported significant lymphoid tissue drug localization with indinavir-associated lipid nanoparticles (LNPs). Our current objective is to evaluate whether additional enhancement is achievable by targeting these particles to CD4-HIV host cells.
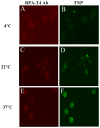
Methods: We characterized 2 peptide-coated (CD4-BP2 and CD4-BP4) drug-associated LNPs and demonstrated CD4-cell specificity. Drug-associated LNPs expressing polyethyleneglycol were exposed on HIV-2-infected cells under dynamic conditions that emulated lymph node physiology for 15, 30, and 60 minutes at concentrations from 0 to 25 μM and evaluated for antiviral activity and cell-associated drug concentrations. The specificity of CD4-mediated enhancement of indinavir LNPs antiviral activity was evaluated by blocking with anti-CD4 antibody.
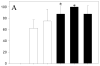
Results: Inclusion of CD4-binding peptides on LNPs enhanced antiviral activity for all incubation conditions, compared with control particles or soluble drug (eg, 60 minutes exposure, EC50 = 0.12-0.13 vs. 0.46 μM for targeted nanoparticles vs. soluble drug). The CD4-BP4 peptide exhibited higher efficiency in eliciting antiviral activity than CD4-BP2-coated particles (EC50 = 7.5 μM vs. >25 μM at 15 minutes drug exposure). This enhancement seems to be driven by CD4 availability and cell-associated indinavir concentrations, as blocking of CD4 significantly ablated indinavir efficacy in targeted particles and indinavir concentrations reflected the observed anti-HIV activity.
Conclusions: We constructed CD4-targeted LNPs that provide selective binding and efficient delivery of indinavir to CD4-HIV host cells. Inclusion of polyethyleneglycol in LNPs would minimize immune recognition of peptides. The enhancement of anti-HIV effects is effective even under limited time exposure.
Figures



Similar articles
-
Optimization of lipid-indinavir complexes for localization in lymphoid tissues of HIV-infected macaques.J Acquir Immune Defic Syndr. 2006 Jun;42(2):155-61. doi: 10.1097/01.qai.0000214822.33905.87. J Acquir Immune Defic Syndr. 2006. PMID: 16760797
-
Anti-HIV drug-combination nanoparticles enhance plasma drug exposure duration as well as triple-drug combination levels in cells within lymph nodes and blood in primates.AIDS Res Hum Retroviruses. 2015 Jan;31(1):107-14. doi: 10.1089/aid.2014.0210. AIDS Res Hum Retroviruses. 2015. PMID: 25402233 Free PMC article.
-
Design and characterization of novel peptide-coated lipid nanoparticles for targeting anti-HIV drug to CD4 expressing cells.AAPS J. 2012 Jun;14(2):225-35. doi: 10.1208/s12248-012-9329-6. Epub 2012 Mar 6. AAPS J. 2012. PMID: 22391788 Free PMC article.
-
Indinavir: a pharmacologic and clinical review of a new HIV protease inhibitor.Conn Med. 1996 Dec;60(12):723-7. Conn Med. 1996. PMID: 9018893 Review. No abstract available.
-
Indinavir: a review of its use in the management of HIV infection.Drugs. 1999 Dec;58(6):1165-203. doi: 10.2165/00003495-199958060-00011. Drugs. 1999. PMID: 10651394 Review.
Cited by
-
Recent developments of nanotherapeutics for targeted and long-acting, combination HIV chemotherapy.Eur J Pharm Biopharm. 2019 May;138:75-91. doi: 10.1016/j.ejpb.2018.04.014. Epub 2018 Apr 17. Eur J Pharm Biopharm. 2019. PMID: 29678735 Free PMC article. Review.
-
Overcoming pharmacologic sanctuaries.Curr Opin HIV AIDS. 2013 May;8(3):190-5. doi: 10.1097/COH.0b013e32835fc68a. Curr Opin HIV AIDS. 2013. PMID: 23454865 Free PMC article. Review.
-
Lipid-calcium phosphate nanoparticles for delivery to the lymphatic system and SPECT/CT imaging of lymph node metastases.Biomaterials. 2014 May;35(16):4688-98. doi: 10.1016/j.biomaterials.2014.02.030. Epub 2014 Mar 6. Biomaterials. 2014. PMID: 24613050 Free PMC article.
-
Long-acting three-drug combination anti-HIV nanoparticles enhance drug exposure in primate plasma and cells within lymph nodes and blood.AIDS. 2014 Nov 13;28(17):2625-7. doi: 10.1097/QAD.0000000000000421. AIDS. 2014. PMID: 25102089 Free PMC article.
-
Nanomedicine based approaches for combating viral infections.J Control Release. 2021 Oct 10;338:80-104. doi: 10.1016/j.jconrel.2021.08.011. Epub 2021 Aug 8. J Control Release. 2021. PMID: 34375690 Free PMC article. Review.
References
-
- Palella FJ, Jr, Delaney KM, Moorman AC, et al. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. HIV Outpatient Study Investigators. The New England journal of medicine. 1998 Mar 26;338(13):853–860. - PubMed
-
- Wong JK, Hezareh M, Gunthard HF, et al. Recovery of replication-competent HIV despite prolonged suppression of plasma viremia. Science. 1997 Nov 14;278(5341):1291–1295. - PubMed
-
- Ruiz L, van Lunzen J, Arno A, et al. Protease inhibitor-containing regimens compared with nucleoside analogues alone in the suppression of persistent HIV-1 replication in lymphoid tissue. Aids. 1999 Jan 14;13(1):F1–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

