The pathways of mitophagy for quality control and clearance of mitochondria
- PMID: 22743996
- PMCID: PMC3524633
- DOI: 10.1038/cdd.2012.81
The pathways of mitophagy for quality control and clearance of mitochondria
Abstract
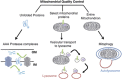
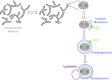
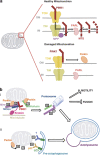
Selective autophagy of mitochondria, known as mitophagy, is an important mitochondrial quality control mechanism that eliminates damaged mitochondria. Mitophagy also mediates removal of mitochondria from developing erythrocytes, and contributes to maternal inheritance of mitochondrial DNA through the elimination of sperm-derived mitochondria. Recent studies have identified specific regulators of mitophagy that ensure selective sequestration of mitochondria as cargo. In yeast, the mitochondrial outer membrane protein autophagy-related gene 32 (ATG32) recruits the autophagic machinery to mitochondria, while mammalian Nix is required for degradation of erythrocyte mitochondria. The elimination of damaged mitochondria in mammals is mediated by a pathway comprised of PTEN-induced putative protein kinase 1 (PINK1) and the E3 ubiquitin ligase Parkin. PINK1 and Parkin accumulate on damaged mitochondria, promote their segregation from the mitochondrial network, and target these organelles for autophagic degradation in a process that requires Parkin-dependent ubiquitination of mitochondrial proteins. Here we will review recent advances in our understanding of the different pathways of mitophagy. In addition, we will discuss the relevance of these pathways in neurons where defects in mitophagy have been implicated in neurodegeneration.
Figures

References
-
- Saraste M. Oxidative phosphorylation at the fin de siecle. Science. 1999;283:1488–1493. - PubMed
-
- Parsons MJ, Green DR. Mitochondria in cell death. Essays Biochem. 2010;47:99–114. - PubMed
-
- Langer T, Kaser M, Klanner C, Leonhard K. AAA proteases of mitochondria: quality control of membrane proteins and regulatory functions during mitochondrial biogenesis. Biochem Soc Trans. 2001;29:431–436. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

