Mechanotransduction in embryonic vascular development
- PMID: 22744845
- PMCID: PMC4502581
- DOI: 10.1007/s10237-012-0412-9
Mechanotransduction in embryonic vascular development
Abstract
A plethora of biochemical signals provides spatial and temporal cues that carefully orchestrate the complex process of vertebrate embryonic development. The embryonic vasculature develops not only in the context of these biochemical cues, but also in the context of the biomechanical forces imparted by blood flow. In the mature vasculature, different blood flow regimes induce distinct genetic programs, and significant progress has been made toward understanding how these forces are perceived by endothelial cells and transduced into biochemical signals. However, it cannot be assumed that paradigms that govern the mature vasculature are pertinent to the developing embryonic vasculature. The embryonic vasculature can respond to the mechanical forces of blood flow, and these responses are critical in vascular remodeling, certain aspects of sprouting angiogenesis, and maintenance of arterial-venous identity. Here, we review data regarding mechanistic aspects of endothelial cell mechanotransduction, with a focus on the response to shear stress, and elaborate upon the multifarious effects of shear stress on the embryonic vasculature. In addition, we discuss emerging predictive vascular growth models and highlight the prospect of combining signaling pathway information with computational modeling. We assert that correlation of precise measurements of hemodynamic parameters with effects on endothelial cell gene expression and cell behavior is required for fully understanding how blood flow-induced loading governs normal vascular development and shapes congenital cardiovascular abnormalities.
Figures

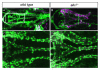

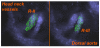
References
-
- Bhullar IS, Li YS, Miao H, Zandi E, Kim M, Shyy JY, Chien S. Fluid shear stress activation of IkappaB kinase is integrin-dependent. J Biol Chem. 1998;273(46):30544–30549. - PubMed
-
- Blum Y, Belting HG, Ellertsdottir E, Herwig L, Luders F, Affolter M. Complex cell rearrangements during intersegmental vessel sprouting and vessel fusion in the zebrafish embryo. Dev Biol. 2008;316(2):312–322. doi:S0012-1606(08)00079-1 [pii] 10.1016/j.ydbio.2008.01.038. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

