Role for NGF in augmented sympathetic nerve response to activation of mechanically and metabolically sensitive muscle afferents in rats with femoral artery occlusion
- PMID: 22744968
- PMCID: PMC3472493
- DOI: 10.1152/japplphysiol.00617.2012
Role for NGF in augmented sympathetic nerve response to activation of mechanically and metabolically sensitive muscle afferents in rats with femoral artery occlusion
Abstract
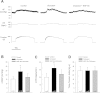
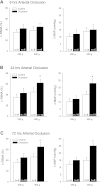
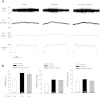
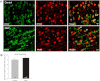
Arterial blood pressure and heart rate responses to static contraction of the hindlimb muscles are greater in rats whose femoral arteries were previously ligated than in control rats. Also, the prior findings demonstrate that nerve growth factor (NGF) is increased in sensory neurons-dorsal root ganglion (DRG) neurons of occluded rats. However, the role for endogenous NGF in engagement of the augmented sympathetic and pressor responses to stimulation of mechanically and/or metabolically sensitive muscle afferent nerves during static contraction after femoral artery ligation has not been specifically determined. In the present study, both afferent nerves and either of them were activated by muscle contraction, passive tendon stretch, and arterial injection of lactic acid into the hindlimb muscles. Data showed that femoral occlusion-augmented blood pressure response to contraction was significantly attenuated by a prior administration of the NGF antibody (NGF-Ab) into the hindlimb muscles. The effects of NGF neutralization were not seen when the sympathetic nerve and pressor responses were evoked by stimulation of mechanically sensitive muscle afferent nerves with tendon stretch in occluded rats. In addition, chemically sensitive muscle afferent nerves were stimulated by lactic acid injected into arterial blood supply of the hindlimb muscles after the prior NGF-Ab, demonstrating that the reflex muscle responses to lactic acid were significantly attenuated. The results of this study further showed that NGF-Ab attenuated an increase in acid-sensing ion channel subtype 3 (ASIC3) of DRG in occluded rats. Moreover, immunohistochemistry was employed to examine the number of C-fiber and A-fiber DRG neurons. The data showed that distribution of DRG neurons with different thin fiber phenotypes was not notably altered when NGF was infused into the hindlimb muscles. However, NGF increased expression of ASIC3 in DRG neurons with C-fiber but not A-fiber. Overall, these data suggest that 1) NGF is amplified in sensory nerves of occluded rats and contributes to augmented reflex sympathetic and blood pressure responses evoked by stimulation of chemically, but not mechanically, sensitive muscle afferent nerves and 2) NGF likely plays a role in modulating the muscle metaboreflex via enhancement of ASIC3 expression in C-fiber of DRG neurons.
Figures



Similar articles
-
Femoral artery occlusion increases expression of ASIC3 in dorsal root ganglion neurons.Am J Physiol Heart Circ Physiol. 2010 Nov;299(5):H1357-64. doi: 10.1152/ajpheart.00612.2010. Epub 2010 Sep 17. Am J Physiol Heart Circ Physiol. 2010. PMID: 20852050 Free PMC article.
-
Contribution of nerve growth factor to upregulation of P2X₃ expression in DRG neurons of rats with femoral artery occlusion.Am J Physiol Heart Circ Physiol. 2011 Sep;301(3):H1070-9. doi: 10.1152/ajpheart.00188.2011. Epub 2011 Jun 3. Am J Physiol Heart Circ Physiol. 2011. PMID: 21642505 Free PMC article.
-
Acid-sensing ion channel subtype 3 function and immunolabelling increases in skeletal muscle sensory neurons following femoral artery occlusion.J Physiol. 2012 Mar 1;590(5):1261-72. doi: 10.1113/jphysiol.2011.221788. Epub 2011 Dec 19. J Physiol. 2012. PMID: 22183722 Free PMC article.
-
Sympathetic Nerve Activity and Blood Pressure Response to Exercise in Peripheral Artery Disease: From Molecular Mechanisms, Human Studies, to Intervention Strategy Development.Int J Mol Sci. 2022 Sep 13;23(18):10622. doi: 10.3390/ijms231810622. Int J Mol Sci. 2022. PMID: 36142521 Free PMC article. Review.
-
Autonomic control of the heart during exercise in humans: role of skeletal muscle afferents.Exp Physiol. 2014 Feb;99(2):300-5. doi: 10.1113/expphysiol.2013.074377. Epub 2013 Aug 30. Exp Physiol. 2014. PMID: 23995102 Review.
Cited by
-
Purinergic P2X Receptors and Heightened Exercise Pressor Reflex in Peripheral Artery Disease.Intern Med Rev (Wash D C). 2016 Nov;2(10):10.18103/imr.v2i10.259. doi: 10.18103/imr.v2i10.259. Intern Med Rev (Wash D C). 2016. PMID: 29862378 Free PMC article.
-
Femoral Artery Occlusion Increases Muscle Pressor Reflex and Expression of Hypoxia-Inducible Factor-1α in Sensory Neurons.J Cardiovasc Dis. 2013 Oct;1(2):34-40. J Cardiovasc Dis. 2013. PMID: 25346936 Free PMC article.
-
TRPA1 mediates amplified sympathetic responsiveness to activation of metabolically sensitive muscle afferents in rats with femoral artery occlusion.Front Physiol. 2015 Sep 15;6:249. doi: 10.3389/fphys.2015.00249. eCollection 2015. Front Physiol. 2015. PMID: 26441669 Free PMC article.
-
Exaggerated cardiovascular responses to muscle contraction and tendon stretch in UCD type-2 diabetes mellitus rats.Am J Physiol Heart Circ Physiol. 2019 Aug 1;317(2):H479-H486. doi: 10.1152/ajpheart.00229.2019. Epub 2019 Jul 5. Am J Physiol Heart Circ Physiol. 2019. PMID: 31274351 Free PMC article.
-
The Role Played by Adenosine in Modulating Reflex Sympathetic and Pressor Responses Evoked by Stimulation of TRPV1 in Muscle Afferents.Cell Physiol Biochem. 2016;40(1-2):39-48. doi: 10.1159/000452523. Epub 2016 Nov 14. Cell Physiol Biochem. 2016. PMID: 27842306 Free PMC article.
References
-
- Baccelli G, Reggiani P, Mattioli A, Corbellini E, Garducci S, Catalano M. The exercise pressor reflex and changes in radial arterial pressure and heart rate during walking in patients with arteriosclerosis obliterans. Angiology 50: 361–374, 1999 - PubMed
-
- Bakke EF, Hisdal J, Jorgensen JJ, Kroese A, Stranden E. Blood pressure in patients with intermittent claudication increases continuously during walking. Eur J Vasc Endovasc Surg 33: 20–25, 2007 - PubMed
-
- Campenot RB, MacInnis BL. Retrograde transport of neurotrophins: fact and function. J Neurobiol 58: 217–229, 2004 - PubMed
-
- Critchley JA, Capewell S. Mortality risk reduction associated with smoking cessation in patients with coronary heart disease: a systematic review. JAMA 290: 86–97, 2003 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

