Retinal laser burn-induced neuropathy leads to substance P-dependent loss of ocular immune privilege
- PMID: 22745377
- PMCID: PMC3401345
- DOI: 10.4049/jimmunol.1103264
Retinal laser burn-induced neuropathy leads to substance P-dependent loss of ocular immune privilege
Abstract
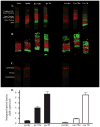
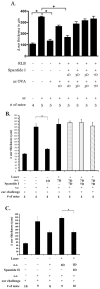
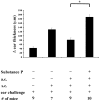
Inflammation in the eye is tightly regulated by multiple mechanisms that together contribute to ocular immune privilege. Many studies have shown that it is very difficult to abrogate the immune privileged mechanism called anterior chamber-associated immune deviation (ACAID). Previously, we showed that retinal laser burn (RLB) to one eye abrogated immune privilege (ACAID) bilaterally for an extended period of time. In an effort to explain the inflammation in the nonburned eye, we postulated that neuronal signals initiated inflammation in the contralateral eye. In this study, we test the role of substance P, a neuroinflamatory peptide, in RLB-induced loss of ACAID. Histological examination of the retina with and without RLB revealed an increase of the substance P-inducible neurokinin 1 receptor (NK1-R) in the retina of first, the burned eye, and then the contralateral eye. Specific antagonists for NK1-R, given locally with Ag within 24 h, but not 3, 5, or 7 d post-RLB treatment, prevented the bilateral loss of ACAID. Substance P knockout (KO) mice retained their ability to develop ACAID post-RLB. These data support the postulate that substance P transmits early inflammatory signals from the RLB eye to the contralateral eye to induce changes to ocular immune privilege and has a central role in the bilateral loss of ACAID. The possibility is raised that blocking of the substance P pathway with NK1-R antagonists postocular trauma may prevent unwanted and perhaps extended consequences of trauma-induced inflammation in the eye.
Figures

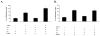
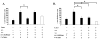
Similar articles
-
Cellular and Molecular Mechanisms of Anterior Chamber-Associated Immune Deviation (ACAID): What We Have Learned from Knockout Mice.Front Immunol. 2017 Nov 30;8:1686. doi: 10.3389/fimmu.2017.01686. eCollection 2017. Front Immunol. 2017. PMID: 29250068 Free PMC article. Review.
-
Retinal laser burn disrupts immune privilege in the eye.Am J Pathol. 2009 Feb;174(2):414-22. doi: 10.2353/ajpath.2009.080766. Epub 2009 Jan 15. Am J Pathol. 2009. PMID: 19147817 Free PMC article.
-
Ocular immune privilege promoted by the presentation of peptide on tolerogenic B cells in the spleen. II. Evidence for presentation by Qa-1.J Immunol. 2001 Jan 1;166(1):26-32. doi: 10.4049/jimmunol.166.1.26. J Immunol. 2001. PMID: 11123273
-
Evidence that peritoneal exudate cells cultured with eye-derived fluids are the proximate antigen-presenting cells in immune deviation of the ocular type.J Immunol. 1993 Nov 15;151(10):5162-71. J Immunol. 1993. PMID: 8228216
-
Neural control of ocular immune privilege.Ann N Y Acad Sci. 2000;917:297-306. doi: 10.1111/j.1749-6632.2000.tb05396.x. Ann N Y Acad Sci. 2000. PMID: 11268357 Review.
Cited by
-
Marked Effects of Tachykinin in Myositis Both in the Experimental Side and Contralaterally: Studies on NK-1 Receptor Expressions in an Animal Model.ISRN Inflamm. 2013 Jan 29;2013:907821. doi: 10.1155/2013/907821. eCollection 2013. ISRN Inflamm. 2013. PMID: 24049666 Free PMC article.
-
Neurokinin-1 Receptor Antagonism Ameliorates Dry Eye Disease by Inhibiting Antigen-Presenting Cell Maturation and T Helper 17 Cell Activation.Am J Pathol. 2020 Jan;190(1):125-133. doi: 10.1016/j.ajpath.2019.09.020. Epub 2019 Oct 24. Am J Pathol. 2020. PMID: 31669306 Free PMC article.
-
The Eye Sees Eye to Eye With the Immune System: The 2019 Proctor Lecture.Invest Ophthalmol Vis Sci. 2019 Oct 1;60(13):4489-4495. doi: 10.1167/iovs.19-28632. Invest Ophthalmol Vis Sci. 2019. PMID: 31661549 Free PMC article. No abstract available.
-
Cellular and Molecular Mechanisms of Anterior Chamber-Associated Immune Deviation (ACAID): What We Have Learned from Knockout Mice.Front Immunol. 2017 Nov 30;8:1686. doi: 10.3389/fimmu.2017.01686. eCollection 2017. Front Immunol. 2017. PMID: 29250068 Free PMC article. Review.
-
The ocular surface immune system through the eyes of aging.Ocul Surf. 2021 Apr;20:139-162. doi: 10.1016/j.jtos.2021.02.007. Epub 2021 Feb 20. Ocul Surf. 2021. PMID: 33621658 Free PMC article. Review.
References
-
- Taylor AW. Neuroimmunomodulation and immune privilege: the role of neuropeptides in ocular immunosuppression. Neuroimmunomodulation. 2002;10:189–198. - PubMed
-
- Maggi CA. The effects of tachykinins on inflammatory and immune cells. Regul Pept. 1997;70:75–90. - PubMed
-
- O'Connor TM, O'Connell J, O'Brien DI, Goode T, Bredin CP, Shanahan F. The role of substance P in inflammatory disease. J Cell Physiol. 2004;201:167–180. - PubMed
-
- Douglas SD, Lai JP, Tuluc F, Schwartz L, Kilpatrick LE. Neurokinin-1 receptor expression and function in human macrophages and brain: perspective on the role in HIV neuropathogenesis. Ann N Y Acad Sci. 2008;1144:90–96. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

