Mitochondrial roles and cytoprotection in chronic liver injury
- PMID: 22745910
- PMCID: PMC3382253
- DOI: 10.1155/2012/387626
Mitochondrial roles and cytoprotection in chronic liver injury
Abstract
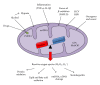
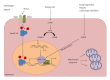
The liver is one of the richest organs in terms of number and density of mitochondria. Most chronic liver diseases are associated with the accumulation of damaged mitochondria. Hepatic mitochondria have unique features compared to other organs' mitochondria, since they are the hub that integrates hepatic metabolism of carbohydrates, lipids and proteins. Mitochondria are also essential in hepatocyte survival as mediator of apoptosis and necrosis. Hepatocytes have developed different mechanisms to keep mitochondrial integrity or to prevent the effects of mitochondrial lesions, in particular regulating organelle biogenesis and degradation. In this paper, we will focus on the role of mitochondria in liver physiology, such as hepatic metabolism, reactive oxygen species homeostasis and cell survival. We will also focus on chronic liver pathologies, especially those linked to alcohol, virus, drugs or metabolic syndrome and we will discuss how mitochondria could provide a promising therapeutic target in these contexts.
Figures
References
-
- Kroemer G, Galluzzi L, Brenner C. Mitochondrial membrane permeabilization in cell death. Physiological Reviews. 2007;87(1):99–163. - PubMed
-
- Veltri KL, Espiritu M, Singh G. Distinct genomic copy number in mitochondria of different mammalian organs. Journal of Cellular Physiology. 1990;143(1):160–164. - PubMed
LinkOut - more resources
Full Text Sources

