Regionally-specific microglial activation in young mice over-expressing human wildtype alpha-synuclein
- PMID: 22750327
- PMCID: PMC3443323
- DOI: 10.1016/j.expneurol.2012.06.025
Regionally-specific microglial activation in young mice over-expressing human wildtype alpha-synuclein
Abstract
Parkinson's disease (PD) is characterized by widespread alpha-synuclein pathology and neuronal loss, primarily of the nigrostriatal dopaminergic neurons. Inflammation has been implicated in PD, and alpha-synuclein can initiate microglial activation; however, the kinetics and distribution of inflammatory responses to alpha-synuclein overexpression in vivo are not well understood. We have examined the regional and temporal pattern of microglial activation and pro-inflammatory cytokine production in mice over-expressing wild-type human alpha-synuclein driven by the Thy1-promoter (Thy1-aSyn mice). An increased number of activated microglia, and increased levels of TNF-α mRNA and protein were first detected in the striatum (1 month of age) and later in the substantia nigra (5-6 months), but not the cerebral cortex or cerebellum; in contrast, IL-1β and TGF-β remained unchanged in the striatum and substantia nigra at all ages examined. Microglial activation persisted up to 14 months of age in these regions and only minimal increases were observed in other regions at this later age. Increased concentrations of serum TNF-α were observed at 5-6 months, but not at 1 month of age. The expression of toll-like receptors (TLRs) 1, TLR 4 and TLR 8, which are possible mediators of microglial activation, was increased at 5-6 months in the substantia nigra but not in the cerebral cortex, and TLR 2 was increased in the substantia nigra at 14 months of age. With the exception of a slight increase in the striatum of 14 month old Thy1-aSyn mice, MHCII staining was not detected in the regions and ages examined. Similarly, peripheral CD4 and CD8-postive T cells were increased in the blood but only at 22 months of age, suggesting later involvement of the adaptive immune response. These data indicate that, despite the presence of high levels of alpha-synuclein in other brain regions, alpha-synuclein overexpression caused a selective early inflammatory response in regions containing the axon terminals and cell bodies of the nigrostriatal pathway. Our results suggest that specific factors, possibly involving a regionally and temporally selective increase in TLRs, mediate alpha-synuclein-induced inflammatory responses in the SN, and may play a role in the selective vulnerability of nigrostriatal dopaminergic neurons in PD.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures
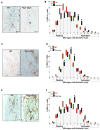
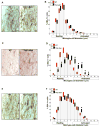
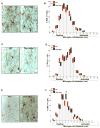




Similar articles
-
Neuroinflammation and protein pathology in Parkinson's disease dementia.Acta Neuropathol Commun. 2020 Dec 3;8(1):211. doi: 10.1186/s40478-020-01083-5. Acta Neuropathol Commun. 2020. PMID: 33272323 Free PMC article.
-
T cell infiltration and upregulation of MHCII in microglia leads to accelerated neuronal loss in an α-synuclein rat model of Parkinson's disease.J Neuroinflammation. 2020 Aug 15;17(1):242. doi: 10.1186/s12974-020-01911-4. J Neuroinflammation. 2020. PMID: 32799878 Free PMC article.
-
Toll-like receptor expression in the blood and brain of patients and a mouse model of Parkinson's disease.Int J Neuropsychopharmacol. 2014 Dec 7;18(6):pyu103. doi: 10.1093/ijnp/pyu103. Int J Neuropsychopharmacol. 2014. PMID: 25522431 Free PMC article.
-
Targeting Microglial α-Synuclein/TLRs/NF-kappaB/NLRP3 Inflammasome Axis in Parkinson's Disease.Front Immunol. 2021 Oct 8;12:719807. doi: 10.3389/fimmu.2021.719807. eCollection 2021. Front Immunol. 2021. PMID: 34691027 Free PMC article. Review.
-
The role of Toll-like receptors and neuroinflammation in Parkinson's disease.J Neuroinflammation. 2022 Jun 6;19(1):135. doi: 10.1186/s12974-022-02496-w. J Neuroinflammation. 2022. PMID: 35668422 Free PMC article. Review.
Cited by
-
Neurons and Glia Interplay in α-Synucleinopathies.Int J Mol Sci. 2021 May 8;22(9):4994. doi: 10.3390/ijms22094994. Int J Mol Sci. 2021. PMID: 34066733 Free PMC article. Review.
-
Relationship among α‑synuclein, aging and inflammation in Parkinson's disease (Review).Exp Ther Med. 2023 Nov 21;27(1):23. doi: 10.3892/etm.2023.12311. eCollection 2024 Jan. Exp Ther Med. 2023. PMID: 38125364 Free PMC article. Review.
-
Increased amoeboid microglial density in the olfactory bulb of Parkinson's and Alzheimer's patients.Brain Pathol. 2014 Mar;24(2):152-65. doi: 10.1111/bpa.12088. Epub 2013 Oct 28. Brain Pathol. 2014. PMID: 24033473 Free PMC article.
-
Complex deregulation and expression of cytokines and mediators of the immune response in Parkinson's disease brain is region dependent.Brain Pathol. 2014 Nov;24(6):584-98. doi: 10.1111/bpa.12137. Epub 2014 Apr 14. Brain Pathol. 2014. PMID: 24593806 Free PMC article.
-
Alpha-Synuclein as a Prominent Actor in the Inflammatory Synaptopathy of Parkinson's Disease.Int J Mol Sci. 2021 Jun 17;22(12):6517. doi: 10.3390/ijms22126517. Int J Mol Sci. 2021. PMID: 34204581 Free PMC article. Review.
References
-
- Alvarez-Erviti L, Couch Y, Richardson J, Cooper JM, Wood MJ. Alpha-synuclein release by neurons activates the inflammatory response in a microglial cell line. J Neurosci Res. 2011;69:337–342. - PubMed
-
- Baba Y, Kuroiwa A, Uitti RJ, Wszolek ZK, Yamada T. Alterations of T-lymphocyte populations in Parkinson disease. Parkinsonism Relat Disord. 2005;11:493–498. - PubMed
-
- Bartels AL, Leenders KL. Neuroinflammation in the pathophysiology of Parkinson’s disease: evidence from animal models to human in vivo studies with [11C]-PK11195 PET. Mov Disord. 2007;22:1852–1856. - PubMed
-
- Batchelor PE, Liberatore GT, Wong JY, Porritt MJ, Frerichs F, Donnan GA, Howells DW. Activated macrophages and microglia induce dopaminergic sprouting in the injured striatum and express brain-derived neurotrophic factor and glial cell line-derived neurotrophic factor. J Neurosci. 1999;19:1708–1716. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

