Memory signals are temporally dissociated in and across human hippocampus and perirhinal cortex
- PMID: 22751037
- PMCID: PMC3428860
- DOI: 10.1038/nn.3154
Memory signals are temporally dissociated in and across human hippocampus and perirhinal cortex
Abstract
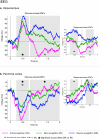
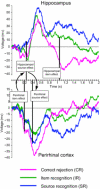
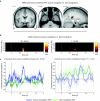
In the endeavor to understand how our brains enable our multifaceted memories, much controversy surrounds the contributions of the hippocampus and perirhinal cortex (PrC). We recorded functional magnetic resonance imaging (fMRI) in healthy controls and intracranial electroencephalography (EEG) in patients during a recognition memory task. Although conventional fMRI analysis showed indistinguishable roles of the hippocampus and PrC in familiarity-based item recognition and recollection-based source retrieval, event-related fMRI and EEG time courses revealed a clear temporal dissociation of memory signals in and across these regions. An early source retrieval effect was followed by a late, post-decision item novelty effect in hippocampus, whereas an early item novelty effect was followed by a sustained source retrieval effect in PrC. Although factors such as memory strength were not experimentally controlled, the temporal pattern across regions suggests that a rapid item recognition signal in PrC triggers a source retrieval process in the hippocampus, which in turn recruits PrC representations and/or mechanisms, evidenced here by increased hippocampal-PrC coupling during source recognition.
Figures


Similar articles
-
Perirhinal cortex supports encoding and familiarity-based recognition of novel associations.Neuron. 2008 Aug 28;59(4):554-60. doi: 10.1016/j.neuron.2008.07.035. Neuron. 2008. PMID: 18760692
-
Perirhinal and parahippocampal cortices differentially contribute to later recollection of object- and scene-related event details.J Neurosci. 2011 Jun 15;31(24):8739-47. doi: 10.1523/JNEUROSCI.4978-10.2011. J Neurosci. 2011. PMID: 21677158 Free PMC article.
-
Medial temporal lobe activity during source retrieval reflects information type, not memory strength.J Cogn Neurosci. 2010 Aug;22(8):1808-18. doi: 10.1162/jocn.2009.21335. J Cogn Neurosci. 2010. PMID: 19702458 Free PMC article.
-
The role of the human hippocampus in familiarity-based and recollection-based recognition memory.Behav Brain Res. 2010 Dec 31;215(2):197-208. doi: 10.1016/j.bbr.2010.04.020. Epub 2010 Apr 20. Behav Brain Res. 2010. PMID: 20412819 Free PMC article. Review.
-
The role of recollection and familiarity in the functional differentiation of the medial temporal lobes.Hippocampus. 2010 Nov;20(11):1291-314. doi: 10.1002/hipo.20853. Hippocampus. 2010. PMID: 20928828 Review.
Cited by
-
Distinct familiarity-based response patterns for faces and buildings in perirhinal and parahippocampal cortex.J Neurosci. 2013 Jun 26;33(26):10915-23. doi: 10.1523/JNEUROSCI.0126-13.2013. J Neurosci. 2013. PMID: 23804111 Free PMC article.
-
The time course of episodic associative retrieval: electrophysiological correlates of cued recall of unimodal and crossmodal pair-associate learning.Cogn Affect Behav Neurosci. 2014 Mar;14(1):220-35. doi: 10.3758/s13415-013-0199-x. Cogn Affect Behav Neurosci. 2014. PMID: 23918600
-
What's the difference between a tiger and a cat? From visual object to semantic concept via the perirhinal cortex.J Neurosci. 2014 Aug 6;34(32):10462-4. doi: 10.1523/JNEUROSCI.2248-14.2014. J Neurosci. 2014. PMID: 25100581 Free PMC article. No abstract available.
-
Functional Specialization of the Medial Temporal Lobes in Human Recognition Memory: Dissociating Effects of Hippocampal versus Parahippocampal Damage.Cereb Cortex. 2022 Apr 5;32(8):1637-1652. doi: 10.1093/cercor/bhab290. Cereb Cortex. 2022. PMID: 34535797 Free PMC article.
-
The Effects of External Cue Overlap and Internal Goals on Selective Memory Retrieval as Revealed by Electroencephalographic (EEG) Neural Pattern Reinstatement.Eur J Neurosci. 2025 Jul;62(1):e70194. doi: 10.1111/ejn.70194. Eur J Neurosci. 2025. PMID: 40653318 Free PMC article.
References
-
- Squire LR, Stark CE, Clark RE. The medial temporal lobe. Annual review of neuroscience. 2004;27:279–306. - PubMed
-
- Davachi L. Item, context and relational episodic encoding in humans. Current opinion in neurobiology. 2006;16:693–700. - PubMed
-
- Mayes A, Montaldi D, Migo E. Associative memory and the medial temporal lobes. Trends Cogn Sci. 2007;11:126–135. - PubMed
-
- Johnson MK, Hashtroudi S, Lindsay DS. Source monitoring. Psychological bulletin. 1993;114:3. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

