Peculiar H⁺ homeostasis of Saccharomyces cerevisiae during the late stages of wine fermentation
- PMID: 22752170
- PMCID: PMC3416607
- DOI: 10.1128/AEM.01355-12
Peculiar H⁺ homeostasis of Saccharomyces cerevisiae during the late stages of wine fermentation
Abstract
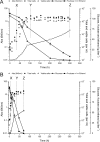
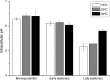
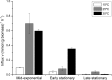
Intracellular pH (pH(in)) is a tightly regulated physiological parameter, which controls cell performance in all living systems. The purpose of this work was to evaluate if and how H(+) homeostasis is accomplished by an industrial wine strain of Saccharomyces cerevisiae while fermenting real must under the harsh winery conditions prevalent in the late stages of the fermentation process, in particular low pH and high ethanol concentrations and temperature. Cells grown at 15, 25, and 30°C were harvested in exponential and early and late stationary phases. Intracellular pH remained in the range of 6.0 to 6.4, decreasing significantly only by the end of glucose fermentation, in particular at lower temperatures (pH(in) 5.2 at 15°C), although the cells remained viable and metabolically active. The cell capability of extruding H(+) via H(+)-ATPase and of keeping H(+) out by means of an impermeable membrane were evaluated as potential mechanisms of H(+) homeostasis. At 30°C, H(+) efflux was higher in all stages. The most striking observation was that cells in late stationary phase became almost impermeable to H(+). Even when these cells were challenged with high ethanol concentrations (up to 20%) added in the assay, their permeability to H(+) remained very low, being almost undetectable at 15°C. Comparatively, ethanol significantly increased the H(+) permeability of cells in exponential phase. Understanding the molecular and physiological events underlying yeast H(+) homeostasis at late stages of fermentations may contribute to the development of more robust strains suitable to efficiently produce a high-quality wine.
Figures


References
-
- Aguilera F, Peinado RA, Millán C, Ortega JM, Mauricio JC. 2006. Relationship between ethanol tolerance, H+-ATPase activity and the lipid composition of the plasma membrane in different wine yeast strains. Int. J. Food Microbiol. 110: 34–42 - PubMed
-
- D'Amore T, Panchal CJ, Russell I, Stewart GG. 1990. A study of ethanol tolerance in yeast. Crit. Rev. Biotechnol. 9: 287–304 - PubMed
-
- Ding J, et al. 2009. Tolerance and stress response to ethanol in the yeast Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 85: 253–263 - PubMed
-
- Fujita K, Matsuyama A, Kobayashi Y, Iwahashi H. 2006. The genome-wide screening of yeast deletion mutants to identify the genes required for tolerance to ethanol and other alcohols. FEMS Yeast Res. 6: 744–750 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

