Conserved residues of the putative L6 loop of Escherichia coli BamA play a critical role in the assembly of β-barrel outer membrane proteins, including that of BamA itself
- PMID: 22753067
- PMCID: PMC3415531
- DOI: 10.1128/JB.00825-12
Conserved residues of the putative L6 loop of Escherichia coli BamA play a critical role in the assembly of β-barrel outer membrane proteins, including that of BamA itself
Abstract
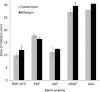
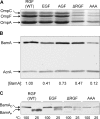
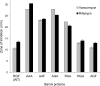
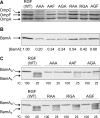
Many members of the Omp85 family of proteins form essential β-barrel outer membrane protein (OMP) biogenesis machinery in Gram-negative bacteria, chloroplasts, and mitochondria. In Escherichia coli, BamA, a member of the Omp85 family, folds into an outer membrane-embedded β-barrel domain and a soluble periplasmic polypeptide-transport-associated (POTRA) domain. Although the high-resolution structures of only the BamA POTRA domain of E. coli are available, the crystal structure of FhaC, an Omp85 family member and a component of the two-partner secretion system in Bordetella pertussis, suggests that the BamA β-barrel likely folds into a 16-stranded β-barrel. The FhaC β-barrel is occluded by an N-terminal α-helix and a large β-barrel loop, L6, which carries residues that are highly conserved among the Omp85 family members. Deletion of L6 in FhaC did not affect its biogenesis but abolished its secretion function. In this study, we tested the hypothesis that the conserved residues of the putative L6 loop, which presumably folds back into the lumen of the BamA β-barrel like the FhaC counterpart, play an important role in OMP and/or BamA biogenesis. The conserved (641)RGF(643) residues of L6 were either deleted or replaced with alanine in various permutations. Phenotypic and biochemical characterization of various BamA L6 mutants revealed that the conserved RGF residues are critical for OMP biogenesis. Moreover, three BamA L6 alterations, ΔRGF, AAA, and AGA, produced a conditional lethal phenotype, concomitant with severely reduced BamA levels and folding defects. Thus, the conserved (641)RGF(643) residues of the BamA L6 loop are important for BamA folding and biogenesis.
Figures



Similar articles
-
Substitutions in the BamA β-barrel domain overcome the conditional lethal phenotype of a ΔbamB ΔbamE strain of Escherichia coli.J Bacteriol. 2012 Jan;194(2):317-24. doi: 10.1128/JB.06192-11. Epub 2011 Oct 28. J Bacteriol. 2012. PMID: 22037403 Free PMC article.
-
Genetic, biochemical, and molecular characterization of the polypeptide transport-associated domain of Escherichia coli BamA.J Bacteriol. 2012 Jul;194(13):3512-21. doi: 10.1128/JB.06740-11. Epub 2012 Apr 27. J Bacteriol. 2012. PMID: 22544271 Free PMC article.
-
Conformation-specific labeling of BamA and suppressor analysis suggest a cyclic mechanism for β-barrel assembly in Escherichia coli.Proc Natl Acad Sci U S A. 2013 Mar 26;110(13):5151-6. doi: 10.1073/pnas.1302662110. Epub 2013 Mar 11. Proc Natl Acad Sci U S A. 2013. PMID: 23479609 Free PMC article.
-
Augmenting β-augmentation: structural basis of how BamB binds BamA and may support folding of outer membrane proteins.J Mol Biol. 2011 Mar 11;406(5):659-66. doi: 10.1016/j.jmb.2011.01.002. Epub 2011 Jan 12. J Mol Biol. 2011. PMID: 21236263 Review.
-
The β-Barrel Assembly Machinery Complex.Methods Mol Biol. 2015;1329:1-16. doi: 10.1007/978-1-4939-2871-2_1. Methods Mol Biol. 2015. PMID: 26427672 Review.
Cited by
-
Receptor polymorphism restricts contact-dependent growth inhibition to members of the same species.mBio. 2013 Jul 23;4(4):e00480-13. doi: 10.1128/mBio.00480-13. mBio. 2013. PMID: 23882017 Free PMC article.
-
How the assembly and protection of the bacterial cell envelope depend on cysteine residues.J Biol Chem. 2020 Aug 21;295(34):11984-11994. doi: 10.1074/jbc.REV120.011201. Epub 2020 Jun 2. J Biol Chem. 2020. PMID: 32487747 Free PMC article. Review.
-
Structural insight into the biogenesis of β-barrel membrane proteins.Nature. 2013 Sep 19;501(7467):385-90. doi: 10.1038/nature12521. Epub 2013 Sep 1. Nature. 2013. PMID: 23995689 Free PMC article.
-
Plasticity within the barrel domain of BamA mediates a hybrid-barrel mechanism by BAM.Nat Commun. 2021 Dec 8;12(1):7131. doi: 10.1038/s41467-021-27449-4. Nat Commun. 2021. PMID: 34880256 Free PMC article.
-
Monoclonal antibody targeting the β-barrel assembly machine of Escherichia coli is bactericidal.Proc Natl Acad Sci U S A. 2018 Apr 3;115(14):3692-3697. doi: 10.1073/pnas.1800043115. Epub 2018 Mar 19. Proc Natl Acad Sci U S A. 2018. PMID: 29555747 Free PMC article.
References
-
- Andrès C, Agne B, Kessler F. 2010. The TOC complex: preprotein gateway to the chloroplast. Biochim. Biophys. Acta 1803:715–723 - PubMed
-
- Becker T, Böttinger L, Pfanner N. 2012. Mitochondrial protein import: from transport pathways to an integrated network. Trends Biochem. Sci. 37:85–91 - PubMed
-
- Casadaban MJ. 1976. Transposition and fusion of the lac genes to selected promoters in Escherichia coli using bacteriophage lambda and Mu. J. Mol. Biol. 104:541–555 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

