Sulfonation of glycopeptide antibiotics by sulfotransferase StaL depends on conformational flexibility of aglycone scaffold
- PMID: 22753479
- PMCID: PMC3406828
- DOI: 10.1073/pnas.1205377109
Sulfonation of glycopeptide antibiotics by sulfotransferase StaL depends on conformational flexibility of aglycone scaffold
Abstract
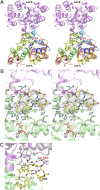
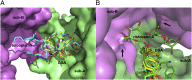
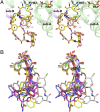
Although glycopeptide antibiotics (GPAs), including vancomycin and teicoplanin, represent the most important class of anti-infective agents in the treatment of serious gram-positive bacterial infections, their usefulness is threatened by the emergence of resistant strains. GPAs are complex natural products consisting of a heptapeptide skeleton assembled via nonribosomal peptide synthesis and constrained through multiple crosslinks, with diversity resulting from enzymatic modifications by a variety of tailoring enzymes, which can be used to produce GPA analogues that could overcome antibiotic resistance. GPA-modifying sulfotransferases are promising tools for generating the unique derivatives. Despite significant sequence and structural similarities, these sulfotransferases modify distinct side chains on the GPA scaffold. To provide insight into the spatial diversity of modifications, we have determined the crystal structure of the ternary complex of bacterial sulfotransferase StaL with the cofactor product 3'-phosphoadenosine 5'-phosphate and desulfo-A47934 aglycone substrate. Desulfo-A47934 binds with the hydroxyl group on the 4-hydroxyphenylglycine in residue 1 directed toward the 3'-phosphoadenosine 5'-phosphate and hydrogen-bonded to the catalytic His67. Homodimeric StaL can accommodate GPA substrate in only one of the two active sites because of potential steric clashes. Importantly, the aglycone substrate demonstrates a flattened conformation, in contrast to the cup-shaped structures observed previously. Analysis of the conformations of this scaffold showed that despite the apparent rigidity due to crosslinking between the side chains, the aglycone scaffold displays substantial flexibility, important for enzymatic modifications by the GPA-tailoring enzymes. We also discuss the potential of using the current structural information in generating unique GPA derivatives.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Crystal structure of StaL, a glycopeptide antibiotic sulfotransferase from Streptomyces toyocaensis.J Biol Chem. 2007 Apr 27;282(17):13073-86. doi: 10.1074/jbc.M611912200. Epub 2007 Feb 28. J Biol Chem. 2007. PMID: 17329243
-
Biosynthesis of sulfated glycopeptide antibiotics by using the sulfotransferase StaL.Chem Biol. 2006 Feb;13(2):171-81. doi: 10.1016/j.chembiol.2005.12.003. Chem Biol. 2006. PMID: 16492565
-
Crystal structures of the glycopeptide sulfotransferase Teg12 in a complex with the teicoplanin aglycone.Biochemistry. 2010 May 18;49(19):4159-68. doi: 10.1021/bi100150v. Biochemistry. 2010. PMID: 20361791 Free PMC article.
-
Biological, chemical, and biochemical strategies for modifying glycopeptide antibiotics.J Biol Chem. 2019 Dec 6;294(49):18769-18783. doi: 10.1074/jbc.REV119.006349. Epub 2019 Oct 31. J Biol Chem. 2019. PMID: 31672921 Free PMC article. Review.
-
Crystal structures of human sulfotransferases: insights into the mechanisms of action and substrate selectivity.Expert Opin Drug Metab Toxicol. 2012 Jun;8(6):635-46. doi: 10.1517/17425255.2012.677027. Epub 2012 Apr 19. Expert Opin Drug Metab Toxicol. 2012. PMID: 22512672 Review.
Cited by
-
Opportunities for synthetic biology in antibiotics: expanding glycopeptide chemical diversity.ACS Synth Biol. 2015 Mar 20;4(3):195-206. doi: 10.1021/sb300092n. Epub 2012 Dec 28. ACS Synth Biol. 2015. PMID: 23654249 Free PMC article. Review.
-
Combinatorial Biosynthesis of Sulfated Benzenediol Lactones with a Phenolic Sulfotransferase from Fusarium graminearum PH-1.mSphere. 2020 Nov 25;5(6):e00949-20. doi: 10.1128/mSphere.00949-20. mSphere. 2020. PMID: 33239367 Free PMC article.
-
From Steroid and Drug Metabolism to Glycobiology, Using Sulfotransferase Structures to Understand and Tailor Function.Drug Metab Dispos. 2022 Jul;50(7):1027-1041. doi: 10.1124/dmd.121.000478. Epub 2022 Feb 22. Drug Metab Dispos. 2022. PMID: 35197313 Free PMC article. Review.
References
-
- Kahne D, Leimkuhler C, Lu W, Walsh C. Glycopeptide and lipoglycopeptide antibiotics. Chem Rev. 2005;105:425–448. - PubMed
-
- Courvalin P. Vancomycin resistance in gram-positive cocci. Clin Infect Dis. 2006;42(Suppl 1):S25–S34. - PubMed
-
- Chang MH, Kish TD, Fung HB. Telavancin: A lipoglycopeptide antimicrobial for the treatment of complicated skin and skin structure infections caused by gram-positive bacteria in adults. Clin Ther. 2010;32:2160–2185. - PubMed
-
- Ashford PA, Bew SP. Recent advances in the synthesis of new glycopeptide antibiotics. Chem Soc Rev. 2012;41:957–978. - PubMed
-
- Mulichak AM, Lu W, Losey HC, Walsh CT, Garavito RM. Crystal structure of vancosaminyltransferase GtfD from the vancomycin biosynthetic pathway: Interactions with acceptor and nucleotide ligands. Biochemistry. 2004;43:5170–5180. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

