Cyclothiazide-induced persistent increase in respiratory-related activity in vitro
- PMID: 22753547
- PMCID: PMC3487044
- DOI: 10.1113/jphysiol.2012.232421
Cyclothiazide-induced persistent increase in respiratory-related activity in vitro
Abstract
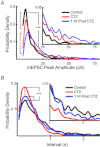
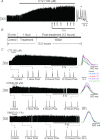
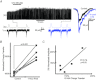
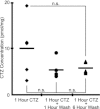
Hypoglossal (XII) motoneurons (MNs) innervate the genioglossus muscle of the tongue, which plays an important role in maintaining upper airway patency, particularly during sleep, and modulating upper airway resistance. Discovering methods for inducing long-term increases in genioglossal motoneuronal excitability to AMPA-mediated drive may help in the development of therapeutics for upper airway motor disorders such as obstructive sleep apnoea. We show that the diuretic, anti-hypertensive, AMPA receptor modulator cyclothiazide (CTZ) induces a profound and long-lasting increase in the amplitude of respiratory-related XII nerve activity in rhythmically active neonatal rat medullary slices. Treatment of the slice with CTZ (90 μM) for 1 h increased the integrated XII ( XII) nerve burst amplitude to 262 ± 23% of pre-treatment control at 1 h post-treatment;much of this increase lasted at least 12 h. The amount of CTZ-induced facilitation (CIF) was dependent upon both CTZ dose and exposure time and was accompanied by a long-lasting increase in endogenous AMPA-mediated drive currents to XII MNs. CIF, however, is not a form of plasticity and does not depend on AMPA or NMDA receptor activation for its induction. Nor does it depend on coincident protein kinase A or C activity. Rather, measurement of mEPSCs along with mass spectrometric analysis of CTZ-treated slices indicates that the cause is prolonged bioavailability of CTZ. These results illustrate a latent residual capacity for potentiating AMPA-mediated inspiratory drive to XII MNs that might be applied to the treatment of upper airway motor deficits.
Figures





Similar articles
-
Modulation of neural network activity in vitro by cyclothiazide, a drug that blocks desensitization of AMPA receptors.J Neurosci. 1995 May;15(5 Pt 2):4046-56. doi: 10.1523/JNEUROSCI.15-05-04046.1995. J Neurosci. 1995. PMID: 7751964 Free PMC article.
-
Cholinergic neurotransmission in the preBötzinger Complex modulates excitability of inspiratory neurons and regulates respiratory rhythm.Neuroscience. 2005;130(4):1069-81. doi: 10.1016/j.neuroscience.2004.10.028. Neuroscience. 2005. PMID: 15653001 Free PMC article.
-
Generation and transmission of respiratory oscillations in medullary slices: role of excitatory amino acids.J Neurophysiol. 1993 Oct;70(4):1497-515. doi: 10.1152/jn.1993.70.4.1497. J Neurophysiol. 1993. PMID: 8283211
-
Noradrenergic modulation of hypoglossal motoneuron excitability: developmental and putative state-dependent mechanisms.Arch Ital Biol. 2011 Dec 1;149(4):426-53. doi: 10.4449/aib.v149i4.1271. Arch Ital Biol. 2011. PMID: 22205594 Review.
-
Modulation of hypoglossal motoneuron excitability by intracellular signal transduction cascades.Respir Physiol Neurobiol. 2005 Jul 28;147(2-3):131-43. doi: 10.1016/j.resp.2005.03.014. Respir Physiol Neurobiol. 2005. PMID: 15893504 Review.
References
-
- Abidin I, Köhler T, Weiler E, Zoidl G, Eysel UT, Lessmann V, Mittmann T. Reduced presynaptic efficiency of excitatory synaptic transmission impairs LTP in the visual cortex of BDNF-heterozygous mice. Eur J Neurosci. 2006;24:3519–3531. - PubMed
-
- Arai AC, Kessler M. Pharmacology of ampakine modulators: from AMPA receptors to synapses and behavior. Curr Drug Targets. 2007;8:583–602. - PubMed
-
- Ballerini L, Bracci E, Nistri A. Desensitization of AMPA receptors limits the amplitude of EPSPs and the excitability of motoneurons of the rat isolated spinal cord. Eur J Neurosci. 1995;7:1229–1234. - PubMed
-
- Bertolino M, Bareli M, Parenti C, Braghiroli D, DiBella M, Vicini S, Costa E. Modulation of AMPA/kainate receptors by analogues of diazoxide and cyclothiazide in thin slices of rat hippocampus. Receptors Channels. 1993;1:267–278. - PubMed
-
- Black MD. Therapeutic potential of positive AMPA modulators and their relationship to AMPA receptor subunits. A review of preclinical data. Psychopharmacology (Berl) 2005;179:154–163. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

