Determinants of cell-material crosstalk at the interface: towards engineering of cell instructive materials
- PMID: 22753785
- PMCID: PMC3405766
- DOI: 10.1098/rsif.2012.0308
Determinants of cell-material crosstalk at the interface: towards engineering of cell instructive materials
Abstract
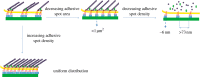
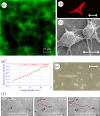
The development of novel biomaterials able to control cell activities and direct their fate is warranted for engineering functional biological tissues, advanced cell culture systems, single-cell diagnosis as well as for cell sorting and differentiation. It is well established that crosstalk at the cell-material interface occurs and this has a profound influence on cell behaviour. However, the complete deciphering of the cell-material communication code is still far away. A variety of material surface properties have been reported to affect the strength and the nature of the cell-material interactions, including biological cues, topography and mechanical properties. Novel experimental evidence bears out the hypothesis that these three different signals participate in the same material-cytoskeleton crosstalk pathway via adhesion plaque formation dynamics. In this review, we present the relevant findings on material-induced cell response along with the description of cell behaviour when exposed to arrays of signals-biochemical, topographical and mechanical. Finally, with the aid of literature data, we attempt to draw unifying elements of the material-cytoskeleton-cell fate chain.
Figures


References
-
- Lehnert D., Wehrle-Haller B., David C., Weiland U., Ballestrem C., Imhof B. A., Bastmeyer M. 2004. Cell behaviour on micropatterned substrata: limits of extracellular matrix geometry for spreading and adhesion. J. Cell Sci. 117, 41–52 10.1242/jcs.00836 (doi:10.1242/jcs.00836) - DOI - PubMed
-
- Flemming R. G., Murphy C. J., Abrams G. A., Goodman S. L., Nealey P. F. 1999. Effects of synthetic micro- and nano-structured surfaces on cell behavior. Biomaterials 20, 573–588 10.1016/S0142-9612(98)00209-9 (doi:10.1016/S0142-9612(98)00209-9) - DOI - PubMed
-
- van Kooten T. G., Spijker H. T., Busscher H. J. 2004. Plasma-treated polystyrene surfaces: model surfaces for studying cell–biomaterial interactions. Biomaterials 25, 1735–1747 10.1016/j.biomaterials.2003.08.071 (doi:10.1016/j.biomaterials.2003.08.071) - DOI - PubMed
-
- Dowling D. P., Miller I. S., Ardhaoui M., Gallagher W. M. 2011. Effect of surface wettability and topography on the adhesion of osteosarcoma cells on plasma-modified polystyrene. J. Biomater. Appl. 26, 327–347 10.1177/0885328210372148 (doi:10.1177/0885328210372148) - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
