Proof of concept for low-dose molecular breast imaging with a dual-head CZT gamma camera. Part I. Evaluation in phantoms
- PMID: 22755726
- PMCID: PMC3376156
- DOI: 10.1118/1.4718665
Proof of concept for low-dose molecular breast imaging with a dual-head CZT gamma camera. Part I. Evaluation in phantoms
Abstract
Purpose: Molecular breast imaging (MBI) is a nuclear medicine technology that uses dual-head cadmium zinc telluride (CZT) gamma cameras to image functional uptake of a radiotracer, Tc-99m sestamibi, in the breast. An important factor in adoption of MBI in the screening setting is reduction of the necessary administered dose of Tc-99m sestamibi from the typically used dose of 740 MBq to approximately 148 MBq, such that MBI's whole-body effective dose is comparable to that of screening mammography. Methods that increase MBI count sensitivity may allow a proportional reduction in the necessary administered dose. Our objective was to evaluate the impact of two count sensitivity improvement methods on image quality by evaluating count sensitivity, spatial resolution, and lesion contrast in phantom simulations.
Methods: Two dual-head CZT-based MBI systems were studied: LumaGem and Discovery NM 750b. Two count sensitivity improvement methods were implemented: registered collimators optimized for dedicated breast imaging and widened energy acceptance window optimized for use with CZT. System sensitivity, spatial resolution, and tumor contrast-to-noise ratio (CNR) were measured comparing standard collimation and energy window setting [126-154 keV (+10%, -10%)] with optimal collimation and a wide energy window [110-154 keV (+10%, -21%)].
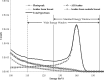
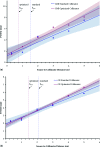
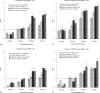
Results: Compared to the standard collimator designs and energy windows for these two systems, use of registered optimized collimation and wide energy window increased system sensitivity by a factor of 2.8-3.6. Spatial resolution decreased slightly for both systems with new collimation. At 3 cm from the collimator face, LumaGem's spatial resolution was 4.8 and 5.6 mm with standard and optimized collimation; Discovery NM 750b's spatial resolution was 4.4 and 4.6 mm with standard and optimized collimation, respectively. For both systems, at tumor depths of 1 and 3 cm, use of optimized collimation and wide energy window significantly improved CNR compared to standard settings for tumors 8.0 and 9.2 mm in diameter. At the closer depth of 1 cm, optimized collimation and wide energy window also significantly improved CNR for 5.9 mm tumors on Discovery NM 750b.
Conclusions: Registered optimized collimation and wide energy window yield a substantial gain in count sensitivity and measurable gain in CNR, with some loss in spatial resolution compared to the standard collimator designs and energy windows used on these two systems. At low-count densities calculated to represent doses of 148 MBq, this tradeoff results in adequate count density and lesion contrast for detection of lesions ≥8 mm in the middle of a typical breast (3 cm deep) and lesions ≥6 mm close to the collimator (1 cm deep).
© 2012 American Association of Physicists in Medicine.
Figures



Similar articles
-
Collimator design for a dedicated molecular breast imaging-guided biopsy system: proof-of-concept.Med Phys. 2013 Jan;40(1):012503. doi: 10.1118/1.4770274. Med Phys. 2013. PMID: 23298114 Free PMC article.
-
Proof of concept for low-dose molecular breast imaging with a dual-head CZT gamma camera. Part II. Evaluation in patients.Med Phys. 2012 Jun;39(6):3476-83. doi: 10.1118/1.4719959. Med Phys. 2012. PMID: 22755727 Free PMC article.
-
Performance characteristics of dedicated molecular breast imaging systems at low doses.Med Phys. 2016 Jun;43(6):3062-3070. doi: 10.1118/1.4950873. Med Phys. 2016. PMID: 27277053
-
Performance of cardiac cadmium-zinc-telluride gamma camera imaging in coronary artery disease: a review from the cardiovascular committee of the European Association of Nuclear Medicine (EANM).Eur J Nucl Med Mol Imaging. 2016 Dec;43(13):2423-2432. doi: 10.1007/s00259-016-3467-5. Epub 2016 Aug 19. Eur J Nucl Med Mol Imaging. 2016. PMID: 27542010 Review.
-
Novel Cadmium Zinc Telluride Devices for Myocardial Perfusion Imaging-Technological Aspects and Clinical Applications.Semin Nucl Med. 2016 Jul;46(4):273-85. doi: 10.1053/j.semnuclmed.2016.01.002. Semin Nucl Med. 2016. PMID: 27237438 Review.
Cited by
-
Direct-Conversion Molecular Breast Imaging of Invasive Breast Cancer: Imaging Features, Extent of Invasive Disease, and Comparison Between Invasive Ductal and Lobular Histology.AJR Am J Roentgenol. 2015 Sep;205(3):W374-81. doi: 10.2214/AJR.14.13502. AJR Am J Roentgenol. 2015. PMID: 26295674 Free PMC article.
-
Collimator design for a dedicated molecular breast imaging-guided biopsy system: proof-of-concept.Med Phys. 2013 Jan;40(1):012503. doi: 10.1118/1.4770274. Med Phys. 2013. PMID: 23298114 Free PMC article.
-
Nuclear imaging of the breast: translating achievements in instrumentation into clinical use.Med Phys. 2013 May;40(5):050901. doi: 10.1118/1.4802733. Med Phys. 2013. PMID: 23635248 Free PMC article. Review.
-
Contrast-noise-ratio (CNR) analysis and optimisation of breast-specific gamma imaging (BSGI) acquisition protocols.EJNMMI Res. 2013 Mar 25;3(1):21. doi: 10.1186/2191-219X-3-21. EJNMMI Res. 2013. PMID: 23531207 Free PMC article.
-
Implementation and evaluation of an expectation maximization reconstruction algorithm for gamma emission breast tomosynthesis.Med Phys. 2012 Dec;39(12):7580-92. doi: 10.1118/1.4764480. Med Phys. 2012. PMID: 23231306 Free PMC article.
References
-
- Kolb T. M., Lichy J., and Newhouse J. H., “Comparison of the performance of screening mammography, physical examination, and breast ultrasound and evaluation of factors that influence them: An analysis of 27,825 patient evaluations,” Radiology 225, 165–175 (2002).10.1148/radiol.2251011667 - DOI - PubMed
-
- Carney P. A., Miglioretti D. L., Yankaskas B. C., Kerlikowske K., Rosenberg R., Rutter C. M., Geller B. M., Abraham L. A., Taplin S. H., Dignan M., Cutter G., and Ballard-Barbash R., “Individual and combined effects of age, breast density, and hormone replacement therapy use on the accuracy of screening mammography,” Ann. Intern Med. 138, 168–175 (2003). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

