Patient characteristics associated with venous thromboembolic events: a cohort study using pooled electronic health record data
- PMID: 22759621
- PMCID: PMC3534456
- DOI: 10.1136/amiajnl-2011-000782
Patient characteristics associated with venous thromboembolic events: a cohort study using pooled electronic health record data
Abstract
Objective: To demonstrate the potential of de-identified clinical data from multiple healthcare systems using different electronic health records (EHR) to be efficiently used for very large retrospective cohort studies.
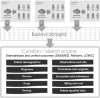
Materials and methods: Data of 959 030 patients, pooled from multiple different healthcare systems with distinct EHR, were obtained. Data were standardized and normalized using common ontologies, searchable through a HIPAA-compliant, patient de-identified web application (Explore; Explorys Inc). Patients were 26 years or older seen in multiple healthcare systems from 1999 to 2011 with data from EHR.
Results: Comparing obese, tall subjects with normal body mass index, short subjects, the venous thromboembolic events (VTE) OR was 1.83 (95% CI 1.76 to 1.91) for women and 1.21 (1.10 to 1.32) for men. Weight had more effect then height on VTE. Compared with Caucasian, Hispanic/Latino subjects had a much lower risk of VTE (female OR 0.47, 0.41 to 0.55; male OR 0.24, 0.20 to 0.28) and African-Americans a substantially higher risk (female OR 1.83, 1.76 to 1.91; male OR 1.58, 1.50 to 1.66). This 13-year retrospective study of almost one million patients was performed over approximately 125 h in 11 weeks, part time by the five authors.
Discussion: As research informatics tools develop and more clinical data become available in EHR, it is important to study and understand unique opportunities for clinical research informatics to transform the scale and resources needed to perform certain types of clinical research.
Conclusions: With the right clinical research informatics tools and EHR data, some types of very large cohort studies can be completed with minimal resources.
Conflict of interest statement
Figures
References
-
- Jha AK, DesRoches CM, Campbell EG, et al. Use of electronic health records in U.S. hospitals. N Engl J Med 2009;360:1628–38 - PubMed
-
- DesRoches CM, Campbell EG, Rao SR, et al. Electronic health records in ambulatory care—a national survey of physicians. N Engl J Med 2008;359:50–60 - PubMed
-
- Blumenthal D, Tavenner M. The “meaningful use” regulation for electronic health records. N Engl J Med 2010;363:501–4 - PubMed
-
- United States Congress Health Insurance Portability and Accountably Act of 1996. 104th Congress, 1st Session. Public Law 104-191. Washington: GPO, 1996. Print.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

