Pediatric invasive pneumococcal disease in the United States in the era of pneumococcal conjugate vaccines
- PMID: 22763632
- PMCID: PMC3416489
- DOI: 10.1128/CMR.00018-12
Pediatric invasive pneumococcal disease in the United States in the era of pneumococcal conjugate vaccines
Abstract
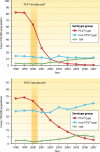
Invasive infections caused by Streptococcus pneumoniae continue to be a major cause of morbidity and mortality worldwide, especially in children under 5 years of age. In the United States, 90% of invasive pneumococcal infections in children are caused by 13 serotypes of S. pneumoniae. The licensure (in 2000) and subsequent widespread use of a heptavalent pneumococcal conjugate vaccine (PCV7) have had a significant impact on decreasing the incidence of serious invasive pneumococcal disease (IPD) in all age groups, especially in children under 2 years of age. However, the emergence of replacement non-PCV7 serotypes, especially serotype 19A, has resulted in an increase in the incidence of serious and invasive infections. In 2010, a 13-valent PCV was licensed in the United States. However, the impact that this vaccine will have on IPD remains to be seen. The objectives of this review are to discuss the epidemiology of serious and invasive pneumococcal infections in the United States in the PCV era and to review some of the pneumococcal vaccines that are in development.
Figures
References
-
- Advisory Committee on Immunization Practices 2000. Preventing pneumococcal diseases among infants and young children. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm. Rep. 49(RR-9):1–35 - PubMed
-
- American Academy of Pediatrics Committee on Infectious Diseases 2000. Recommendations for the prevention of pneumococcal infections, including use of pneumococcal conjugate vaccine (Prevnar), pneumococcal polysaccharide vaccine, and antibiotic prophylaxis. Pediatrics 106:362–366 - PubMed
-
- Arevalo MT, et al. 2009. Mucosal vaccination with a multicomponent adenovirus-vectored vaccine protects against Streptococcus pneumoniae infection in the lung. FEMS Immunol. Med. Microbiol. 55:346–351 - PubMed
-
- Audouy SA, et al. 2007. Development of a lactococcal GEM-based pneumococcal vaccine. Vaccine 25:2497–2506 - PubMed
-
- Baraff LJ, Lee SI, Schriger DL. 1993. Outcomes of bacterial meningitis in children: a meta-analysis. Pediatr. Infect. Dis. J. 12:389–394 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical