Robustness of cortical topography across fields, laminae, anesthetic states, and neurophysiological signal types
- PMID: 22764225
- PMCID: PMC3402176
- DOI: 10.1523/JNEUROSCI.0065-12.2012
Robustness of cortical topography across fields, laminae, anesthetic states, and neurophysiological signal types
Abstract
Topographically organized maps of the sensory receptor epithelia are regarded as cornerstones of cortical organization as well as valuable readouts of diverse biological processes ranging from evolution to neural plasticity. However, maps are most often derived from multiunit activity recorded in the thalamic input layers of anesthetized animals using near-threshold stimuli. Less distinct topography has been described by studies that deviated from the formula above, which brings into question the generality of the principle. Here, we explicitly compared the strength of tonotopic organization at various depths within core and belt regions of the auditory cortex using electrophysiological measurements ranging from single units to delta-band local field potentials (LFP) in the awake and anesthetized mouse. Unit recordings in the middle cortical layers revealed a precise tonotopic organization in core, but not belt, regions of auditory cortex that was similarly robust in awake and anesthetized conditions. In core fields, tonotopy was degraded outside the middle layers or when LFP signals were substituted for unit activity, due to an increasing proportion of recording sites with irregular tuning for pure tones. However, restricting our analysis to clearly defined receptive fields revealed an equivalent tonotopic organization in all layers of the cortical column and for LFP activity ranging from gamma to theta bands. Thus, core fields represent a transition between topographically organized simple receptive field arrangements that extend throughout all layers of the cortical column and the emergence of nontonotopic representations outside the input layers that are further elaborated in the belt fields.
Figures

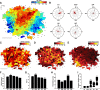
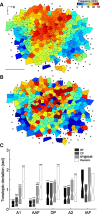
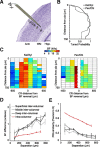


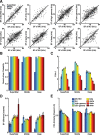

Similar articles
-
Reciprocal modulatory influences between tonotopic and nontonotopic cortical fields in the cat.J Neurosci. 2010 Jan 27;30(4):1476-87. doi: 10.1523/JNEUROSCI.5708-09.2009. J Neurosci. 2010. PMID: 20107075 Free PMC article.
-
Complexity of frequency receptive fields predicts tonotopic variability across species.Elife. 2020 May 18;9:e53462. doi: 10.7554/eLife.53462. Elife. 2020. PMID: 32420865 Free PMC article.
-
Linking topography to tonotopy in the mouse auditory thalamocortical circuit.J Neurosci. 2011 Feb 23;31(8):2983-95. doi: 10.1523/JNEUROSCI.5333-10.2011. J Neurosci. 2011. PMID: 21414920 Free PMC article.
-
Representations in auditory cortex.Curr Opin Neurobiol. 2009 Aug;19(4):430-3. doi: 10.1016/j.conb.2009.07.009. Epub 2009 Aug 10. Curr Opin Neurobiol. 2009. PMID: 19674890 Free PMC article. Review.
-
Local versus global scales of organization in auditory cortex.Trends Neurosci. 2014 Sep;37(9):502-10. doi: 10.1016/j.tins.2014.06.003. Epub 2014 Jul 4. Trends Neurosci. 2014. PMID: 25002236 Free PMC article. Review.
Cited by
-
Encoding sound in the cochlea: from receptor potential to afferent discharge.J Physiol. 2021 May;599(10):2527-2557. doi: 10.1113/JP279189. Epub 2021 Mar 29. J Physiol. 2021. PMID: 33644871 Free PMC article. Review.
-
Persistent Thalamic Sound Processing Despite Profound Cochlear Denervation.Front Neural Circuits. 2016 Aug 31;10:72. doi: 10.3389/fncir.2016.00072. eCollection 2016. Front Neural Circuits. 2016. PMID: 27630546 Free PMC article.
-
Synaptic Basis for the Generation of Response Variation in Auditory Cortex.Sci Rep. 2016 Aug 3;6:31024. doi: 10.1038/srep31024. Sci Rep. 2016. PMID: 27484928 Free PMC article.
-
Contrast gain control in mouse auditory cortex.J Neurophysiol. 2018 Oct 1;120(4):1872-1884. doi: 10.1152/jn.00847.2017. Epub 2018 Jul 25. J Neurophysiol. 2018. PMID: 30044164 Free PMC article.
-
Atypical Response Properties of the Auditory Cortex of Awake MECP2-Overexpressing Mice.Front Neurosci. 2019 May 7;13:439. doi: 10.3389/fnins.2019.00439. eCollection 2019. Front Neurosci. 2019. PMID: 31133783 Free PMC article.
References
-
- Anderson LA, Christianson GB, Linden JF. Mouse auditory cortex differs from visual and somatosensory cortices in the laminar distribution of cytochrome oxidase and acetylcholinesterase. Brain Res. 2009;1252:130–142. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
