Prenatal nicotine exposure mouse model showing hyperactivity, reduced cingulate cortex volume, reduced dopamine turnover, and responsiveness to oral methylphenidate treatment
- PMID: 22764249
- PMCID: PMC3417040
- DOI: 10.1523/JNEUROSCI.1041-12.2012
Prenatal nicotine exposure mouse model showing hyperactivity, reduced cingulate cortex volume, reduced dopamine turnover, and responsiveness to oral methylphenidate treatment
Abstract
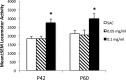
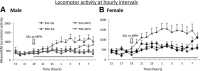
Cigarette smoking, nicotine replacement therapy, and smokeless tobacco use during pregnancy are associated with cognitive disabilities later in life in children exposed prenatally to nicotine. The disabilities include attention deficit hyperactivity disorder (ADHD) and conduct disorder. However, the structural and neurochemical bases of these cognitive deficits remain unclear. Using a mouse model we show that prenatal nicotine exposure produces hyperactivity, selective decreases in cingulate cortical volume, and radial thickness, as well as decreased dopamine turnover in the frontal cortex. The hyperactivity occurs in both male and female offspring and peaks during the "active" or dark phase of the light/dark cycle. These features of the mouse model closely parallel the human ADHD phenotype, whether or not the ADHD is associated with prenatal nicotine exposure. A single oral, but not intraperitoneal, administration of a therapeutic equivalent dose (0.75 mg/kg) of methylphenidate decreases the hyperactivity and increases the dopamine turnover in the frontal cortex of the prenatally nicotine exposed mice, once again paralleling the therapeutic effects of this compound in ADHD subjects. Collectively, our data suggest that the prenatal nicotine exposure mouse model has striking parallels to the ADHD phenotype not only in behavioral, neuroanatomical, and neurochemical features, but also with respect to responsiveness of the behavioral phenotype to methylphenidate treatment. The behavioral, neurochemical, and anatomical biomarkers in the mouse model could be valuable for evaluating new therapies for ADHD and mechanistic investigations into its etiology.
Figures


Similar articles
-
A prenatal nicotine exposure mouse model of methylphenidate responsive ADHD-associated cognitive phenotypes.Int J Dev Neurosci. 2017 May;58:26-34. doi: 10.1016/j.ijdevneu.2017.01.014. Epub 2017 Feb 4. Int J Dev Neurosci. 2017. PMID: 28179105
-
Developmental nicotine exposure precipitates multigenerational maternal transmission of nicotine preference and ADHD-like behavioral, rhythmometric, neuropharmacological, and epigenetic anomalies in adolescent mice.Neuropharmacology. 2019 May 1;149:66-82. doi: 10.1016/j.neuropharm.2019.02.006. Epub 2019 Feb 8. Neuropharmacology. 2019. PMID: 30742847 Free PMC article.
-
Prenatal nicotine exposure decreases the release of dopamine in the medial frontal cortex and induces atomoxetine-responsive neurobehavioral deficits in mice.Psychopharmacology (Berl). 2017 Jun;234(12):1853-1869. doi: 10.1007/s00213-017-4591-z. Epub 2017 Mar 23. Psychopharmacology (Berl). 2017. PMID: 28332006
-
Cingulate impairments in ADHD: Comorbidities, connections, and treatment.Handb Clin Neurol. 2019;166:297-314. doi: 10.1016/B978-0-444-64196-0.00016-9. Handb Clin Neurol. 2019. PMID: 31731917 Review.
-
Prenatal nicotine exposure and child behavioural problems.Eur Child Adolesc Psychiatry. 2014 Oct;23(10):913-29. doi: 10.1007/s00787-014-0615-y. Epub 2014 Sep 21. Eur Child Adolesc Psychiatry. 2014. PMID: 25241028 Free PMC article. Review.
Cited by
-
The impact of drugs of abuse on executive function: characterizing long-term changes in neural correlates following chronic drug exposure and withdrawal in rats.Learn Mem. 2018 Aug 16;25(9):461-473. doi: 10.1101/lm.047001.117. Print 2018 Sep. Learn Mem. 2018. PMID: 30115768 Free PMC article. Review.
-
Effects of the interaction between PTSD and ADHD symptoms on the level of reporting psychotic-like experiences: findings from a non-clinical population.Front Psychiatry. 2023 Oct 6;14:1232606. doi: 10.3389/fpsyt.2023.1232606. eCollection 2023. Front Psychiatry. 2023. PMID: 37867761 Free PMC article.
-
Prenatal Nicotine Exposure Impairs Executive Control Signals in Medial Prefrontal Cortex.Neuropsychopharmacology. 2016 Feb;41(3):716-25. doi: 10.1038/npp.2015.197. Epub 2015 Jul 20. Neuropsychopharmacology. 2016. PMID: 26189451 Free PMC article.
-
Heritable consequences of paternal nicotine exposure: from phenomena to mechanisms†.Biol Reprod. 2021 Sep 14;105(3):632-643. doi: 10.1093/biolre/ioab116. Biol Reprod. 2021. PMID: 34126634 Free PMC article. Review.
-
Transgenerational transmission of hyperactivity in a mouse model of ADHD.J Neurosci. 2014 Feb 19;34(8):2768-73. doi: 10.1523/JNEUROSCI.4402-13.2014. J Neurosci. 2014. PMID: 24553919 Free PMC article.
References
-
- Andersen SL, Arvanitogiannis A, Pliakas AM, LeBlanc C, Carlezon WA., Jr Altered responsiveness to cocaine in rats exposed to methylphenidate during development. Nat Neurosci. 2002;5:13–14. - PubMed
-
- Arnsten AF. Stimulants: Therapeutic Actions in ADHD. Neuropsychopharmacology. 2006;31:2376–2383. - PubMed
-
- Augustyniak PN, Kourrich S, Rezazadeh SM, Stewart J, Arvanitogiannis A. Differential behavioral and neurochemical effects of cocaine after early exposure to methylphenidate in an animal model of attention deficit hyperactivity disorder. Behav Brain Res. 2006;167:379–382. - PubMed
-
- Benowitz NL, Kuyt F, Jacob P., III Circadian blood nicotine concentrations during cigarette smoking. Clin Pharm Ther. 1982;32:758–764. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
