Translational control in cancer etiology
- PMID: 22767671
- PMCID: PMC3552512
- DOI: 10.1101/cshperspect.a012336
Translational control in cancer etiology
Erratum in
- Cold Spring Harb Perspect Biol. 2012 Nov;4(11). doi:10.1101/cshperspect.a015891
Abstract
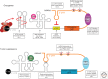
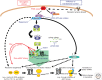
The link between perturbations in translational control and cancer etiology is becoming a primary focus in cancer research. It has now been established that genetic alterations in several components of the translational apparatus underlie spontaneous cancers as well as an entire class of inherited syndromes known as "ribosomopathies" associated with increased cancer susceptibility. These discoveries have illuminated the importance of deregulations in translational control to very specific cellular processes that contribute to cancer etiology. In addition, a growing body of evidence supports the view that deregulation of translational control is a common mechanism by which diverse oncogenic pathways promote cellular transformation and tumor development. Indeed, activation of these key oncogenic pathways induces rapid and dramatic translational reprogramming both by increasing overall protein synthesis and by modulating specific mRNA networks. These translational changes promote cellular transformation, impacting almost every phase of tumor development. This paradigm represents a new frontier in the multihit model of cancer formation and offers significant promise for innovative cancer therapies. Current research, in conjunction with cutting edge technologies, will further enable us to explore novel mechanisms of translational control, functionally identify translationally controlled mRNA groups, and unravel their impact on cellular transformation and tumorigenesis.
Figures

References
-
- Abraham N, Stojdl DF, Duncan PI, Methot N, Ishii T, Dube M, Vanderhyden BC, Atkins HL, Gray DA, McBurney MW, et al. 1999. Characterization of transgenic mice with targeted disruption of the catalytic domain of the double-stranded RNA-dependent protein kinase, PKR. J Biol Chem 274: 5953–5962 - PubMed
-
- Ahlemann M, Zeidler R, Lang S, Mack B, Munz M, Gires O 2006. Carcinoma-associated eIF3i overexpression facilitates mTOR-dependent growth transformation. Mol Carcinog 45: 957–967 - PubMed
-
- Anand N, Murthy S, Amann G, Wernick M, Porter LA, Cukier IH, Collins C, Gray JW, Diebold J, Demetrick DJ, et al. 2002. Protein elongation factor EEF1A2 is a putative oncogene in ovarian cancer. Nat Genet 31: 301–305 - PubMed
-
- Arabi A, Rustum C, Hallberg E, Wright AP 2003. Accumulation of c-Myc and proteasomes at the nucleoli of cells containing elevated c-Myc protein levels. J Cell Sci 116: 1707–1717 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
