PNA bearing 5-azidomethyluracil: a novel approach for solid and solution phase modification
- PMID: 22772040
- PMCID: PMC3429531
- DOI: 10.4161/adna.20158
PNA bearing 5-azidomethyluracil: a novel approach for solid and solution phase modification
Abstract
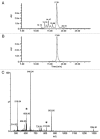
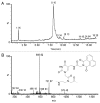
Fmoc- and Boc-protected modified monomers bearing 5-azidomethyluracil nucleobase were synthesized. Four different solid-phase synthetic strategies were tested in order to evaluate the application of this series of monomers for the solid-phase synthesis of modified PNA. The azide was used as masked amine for the introduction of amide-linked functional groups, allowing the production of a library of compounds starting from a single modified monomer. The azide function was also exploited as reactive group for the modification of PNA in solution via azide-alkyne click cycloaddition.
Figures




References
-
- Nielsen PE, Egholm M. Peptide Nucleic Acids: Protocols and Applications. 2. Wymondham: Horizon Press, 2004.
-
- Nielsen PE. Gene targeting using peptide nucleic acid. Methods Mol Biol. 2005;288:343–58. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
