Physical interaction between coat morphogenetic proteins SpoVID and CotE is necessary for spore encasement in Bacillus subtilis
- PMID: 22773792
- PMCID: PMC3430338
- DOI: 10.1128/JB.00914-12
Physical interaction between coat morphogenetic proteins SpoVID and CotE is necessary for spore encasement in Bacillus subtilis
Abstract
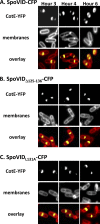
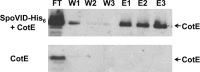
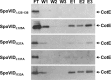
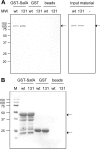
Endospore formation by Bacillus subtilis is a complex and dynamic process. One of the major challenges of sporulation is the assembly of a protective, multilayered, proteinaceous spore coat, composed of at least 70 different proteins. Spore coat formation can be divided into two distinct stages. The first is the recruitment of proteins to the spore surface, dependent on the morphogenetic protein SpoIVA. The second step, known as encasement, involves the migration of the coat proteins around the circumference of the spore in successive waves, a process dependent on the morphogenetic protein SpoVID and the transcriptional regulation of individual coat genes. We provide genetic and biochemical evidence supporting the hypothesis that SpoVID promotes encasement of the spore by establishing direct protein-protein interactions with other coat morphogenetic proteins. It was previously demonstrated that SpoVID directly interacts with SpoIVA and the inner coat morphogenetic protein, SafA. Here, we show by yeast two-hybrid and pulldown assays that SpoVID also interacts directly with the outer coat morphogenetic protein, CotE. Furthermore, by mutational analysis, we identified a specific residue in the N-terminal domain of SpoVID that is essential for the interaction with CotE but dispensable for the interaction with SafA. We propose an updated model of coat assembly and spore encasement that incorporates several physical interactions between the principal coat morphogenetic proteins.
Figures

References
-
- Boland FM, Atrih A, Chirakkal H, Foster SJ, Moir A. 2000. Complete spore-cortex hydrolysis during germination of Bacillus subtilis 168 requires SleB and YpeB. Microbiology 146:57–64 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

