The cell biology of disease: Acute promyelocytic leukemia, arsenic, and PML bodies
- PMID: 22778276
- PMCID: PMC3392943
- DOI: 10.1083/jcb.201112044
The cell biology of disease: Acute promyelocytic leukemia, arsenic, and PML bodies
Abstract
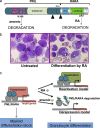
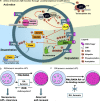
Acute promyelocytic leukemia (APL) is driven by a chromosomal translocation whose product, the PML/retinoic acid (RA) receptor α (RARA) fusion protein, affects both nuclear receptor signaling and PML body assembly. Dissection of APL pathogenesis has led to the rediscovery of PML bodies and revealed their role in cell senescence, disease pathogenesis, and responsiveness to treatment. APL is remarkable because of the fortuitous identification of two clinically effective therapies, RA and arsenic, both of which degrade PML/RARA oncoprotein and, together, cure APL. Analysis of arsenic-induced PML or PML/RARA degradation has implicated oxidative stress in the biogenesis of nuclear bodies and SUMO in their degradation.
Figures


References
-
- Ablain J., Nasr R., Bazarbachi A., de Thé H. 2011. The drug-induced degradation of oncoproteins: an unexpected achilles’ heel of cancer cells? Cancer Discov. 1:117–127 10.1158/2159-8290.CD-11-0087 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

