Identifying the binding site of novel methyllycaconitine (MLA) analogs at α4β2 nicotinic acetylcholine receptors
- PMID: 22778816
- PMCID: PMC3368652
- DOI: 10.1021/cn100073x
Identifying the binding site of novel methyllycaconitine (MLA) analogs at α4β2 nicotinic acetylcholine receptors
Abstract
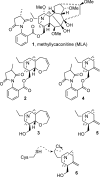
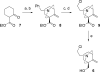
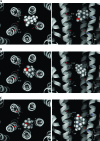
Neuronal nicotinic acetylcholine receptors (nAChR) are ligand gated ion channels that mediate fast synaptic transmission. Methyllycaconitine (MLA) is a selective and potent antagonist of the α7 nAChR, and its anthranilate ester side-chain is important for its activity. Here we report the influence of structure on nAChR inhibition for a series of novel MLA analogs, incorporating either an alcohol or anthranilate ester side-chain to an azabicyclic or azatricyclic core against rat α7, α4β2, and α3β4 nAChRs expressed in Xenopus oocytes. The analogs inhibited ACh (EC(50)) within an IC(50) range of 2.3-26.6 μM. Most displayed noncompetitive antagonism, but the anthranilate ester analogs exerted competitive behavior at the α7 nAChR. At α4β2 nAChRs, inhibition by the azabicyclic alcohol was voltage-dependent suggesting channel block. The channel-lining residues of α4 subunits were mutated to cysteine and the effect of azabicyclic alcohol was evaluated by competition with methanethiosulfonate ethylammonium (MTSEA) and a thiol-reactive probe in the open, closed, and desensitized states of α4β2 nAChRs. The azabicyclic alcohol was found to compete with MTSEA between residues 6' and 13' in a state-dependent manner, but the reactive probe only bonded with 13' in the open state. The data suggest that the 13' position is the dominant binding site. Ligand docking of the azabicyclic alcohol into a (α4)(3)(β2)(2) homology model of the closed channel showed that the ligand can be accommodated at this location. Thus our data reveal distinct pharmacological differences between different nAChR subtypes and also identify a specific binding site for a noncompetitive channel blocker.
Keywords: Ligand-gated ion channel (LGIC); homology model; methyllycaconitine (MLA); nicotinic acetylcholine receptor (nAChR); noncompetitive antagonist (NCA); reactive probe; substituted cysteine accessibility method (SCAM).
Figures
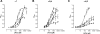
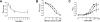
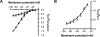

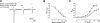



Similar articles
-
Covalent trapping of methyllycaconitine at the α4-α4 interface of the α4β2 nicotinic acetylcholine receptor: antagonist binding site and mode of receptor inhibition revealed.J Biol Chem. 2013 Sep 13;288(37):26521-32. doi: 10.1074/jbc.M113.475053. Epub 2013 Jul 26. J Biol Chem. 2013. PMID: 23893416 Free PMC article.
-
AE Succinimide, an Analogue of Methyllycaconitine, When Bound Generates a Nonconducting Conformation of the α4β2 Nicotinic Acetylcholine Receptor.ACS Chem Neurosci. 2020 Feb 5;11(3):344-355. doi: 10.1021/acschemneuro.9b00525. Epub 2020 Jan 14. ACS Chem Neurosci. 2020. PMID: 31898891
-
Novel methyllycaconitine analogues selective for the α4β2 over α7 nicotinic acetylcholine receptors.Bioorg Med Chem. 2021 Dec 1;51:116516. doi: 10.1016/j.bmc.2021.116516. Epub 2021 Nov 10. Bioorg Med Chem. 2021. PMID: 34798380
-
Nicotinic acetylcholine receptor ligands, a patent review (2006-2011).Expert Opin Ther Pat. 2011 Dec;21(12):1867-96. doi: 10.1517/13543776.2011.637919. Expert Opin Ther Pat. 2011. PMID: 22098319 Free PMC article. Review.
-
Ionotropic and metabotropic responses by alpha 7 nicotinic acetylcholine receptors.Pharmacol Res. 2023 Nov;197:106975. doi: 10.1016/j.phrs.2023.106975. Epub 2023 Oct 29. Pharmacol Res. 2023. PMID: 38032294 Review.
Cited by
-
Covalent trapping of methyllycaconitine at the α4-α4 interface of the α4β2 nicotinic acetylcholine receptor: antagonist binding site and mode of receptor inhibition revealed.J Biol Chem. 2013 Sep 13;288(37):26521-32. doi: 10.1074/jbc.M113.475053. Epub 2013 Jul 26. J Biol Chem. 2013. PMID: 23893416 Free PMC article.
-
NMR structures of the transmembrane domains of the α4β2 nAChR.Biochim Biophys Acta. 2012 May;1818(5):1261-8. doi: 10.1016/j.bbamem.2012.02.008. Epub 2012 Feb 14. Biochim Biophys Acta. 2012. PMID: 22361591 Free PMC article.
-
Structure-Function of Neuronal Nicotinic Acetylcholine Receptor Inhibitors Derived From Natural Toxins.Front Neurosci. 2020 Nov 25;14:609005. doi: 10.3389/fnins.2020.609005. eCollection 2020. Front Neurosci. 2020. PMID: 33324158 Free PMC article. Review.
-
Different Classes of Antidepressants Inhibit the Rat α7 Nicotinic Acetylcholine Receptor by Interacting within the Ion Channel: A Functional and Structural Study.Molecules. 2021 Feb 13;26(4):998. doi: 10.3390/molecules26040998. Molecules. 2021. PMID: 33668529 Free PMC article.
References
-
- Unwin N. (1993) Nicotinic acetylcholine receptor at 9 A resolution. J. Mol. Biol. 229, 1101–1124. - PubMed
-
- Paterson D.; Nordberg A. (2000) Neuronal nicotinic receptors in the human brain. Prog. Neurobiol. 61, 75–111. - PubMed
-
- Romanelli M. N.; Gratteri P.; Guandalini L.; Martini E.; Bonaccini C.; Gualtieri F. (2007) Central nicotinic receptors: Structure, function, ligands, and therapeutic potential. ChemMedChem 2, 746–767. - PubMed
-
- Lukas R. J.; Changeux J. P.; Le Novere N.; Albuquerque E. X.; Balfour D. J.; Berg D. K.; Bertrand D.; Chiappinelli V. A.; Clarke P. B.; Collins A. C.; Dani J. A.; Grady S. R.; Kellar K. J.; Lindstrom J. M.; Marks M. J.; Quik M.; Taylor P. W.; Wonnacott S. (1999) International Union of Pharmacology. XX. Current status of the nomenclature for nicotinic acetylcholine receptors and their subunits. Pharmacol. Rev. 51, 397–401. - PubMed
-
- Anand R.; Conroy W. G.; Schoepfer R.; Whiting P.; Lindstrom J. (1991) Neuronal nicotinic acetylcholine receptors expressed in Xenopus oocytes have a pentameric quaternary structure. J. Biol. Chem. 266, 11192–11198. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

