Delivery of neuropeptides from the periphery to the brain: studies with enkephalin
- PMID: 22778833
- PMCID: PMC3368673
- DOI: 10.1021/cn100001j
Delivery of neuropeptides from the periphery to the brain: studies with enkephalin
Abstract
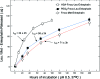
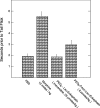
Many peptides with the potential of therapeutic action for brain disorders are not in clinical use because they are unable to cross the blood-brain barrier (BBB) following peripheral administration. We have developed two potential strategies for the delivery of peptides to the brain and demonstrated their feasibility with enkephalins. In the first approach, designated induced reversible lipophilization, Leu/Met Enkephalins were converted to 9-fluorenylmethoxycarbonyl (Fmoc) derived lipophilic prodrug analogues, which undergo slow, spontaneous hydrolysis under physiological conditions, generating the native agonists. In contrast to Enkephalin, Fmoc-Met-Enkephalin was found to facilitate an analgesic effect following intraperitoneal administration in mice. Fmoc-Leu-Enkephalin was not analgesic. In the second approach, Enkephalin was linked to BBB transport vectors through an Fmoc based linker spacer, forming conjugates that slowly release Enkephalin under physiological conditions. A pronounced antinociceptive response was thus obtained following intraperitoneal administration of either cationized-human serum albumin-Fmoc-Enkephalin or polyethylene glycol(5)-Fmoc-Enkephalin. Derivatives of Enkephalin covalently linked to the same BBB-transport vectors through a stable (nonreversible) chemical bond were not analgesic. In summary, we have demonstrated that lipophilicity can be conferred to hydrophilic peptides to a degree permitting the permeation of the BBB by passive diffusion, without the drawback of agonist inactivation, which is often caused by irreversible derivatization. Similarly, in the second strategy, the conjugation to BBB-permeable vectors overcomes the obstacle of peptide inactivation by releasing the active form in the central nervous system.
Keywords: Neuropeptide; blood−brain barrier; enkephalin; prodrug; reversible-conjugation; reversible-lypophilization.
Figures




Similar articles
-
[Effect of ethanol on the concentration of enkephalins in the brain of rats with different levels of alcoholic motivation].Biull Eksp Biol Med. 1983 Jul;96(7):48-51. Biull Eksp Biol Med. 1983. PMID: 6683575 Russian.
-
A prodrug nanoparticle approach for the oral delivery of a hydrophilic peptide, leucine(5)-enkephalin, to the brain.Mol Pharm. 2012 Jun 4;9(6):1665-80. doi: 10.1021/mp300009u. Epub 2012 May 18. Mol Pharm. 2012. PMID: 22574705
-
Nicotine-induced alterations in brain regional concentrations of native and cryptic Met- and Leu-enkephalin.Peptides. 1987 Nov-Dec;8(6):1035-43. doi: 10.1016/0196-9781(87)90133-1. Peptides. 1987. PMID: 3441443
-
Endogenous signaling complexity in neuropeptides- leucine- and methionine-enkephalin.Cell Mol Neurobiol. 2006 Jul-Aug;26(4-6):1003-10. doi: 10.1007/s10571-006-9100-6. Epub 2006 Jun 27. Cell Mol Neurobiol. 2006. PMID: 16802191 Free PMC article. Review.
-
Enkephalins, brain and immunity: modulation of immune responses by methionine-enkephalin injected into the cerebral cavity.Int J Neurosci. 1992 Nov-Dec;67(1-4):241-70. doi: 10.3109/00207459208994788. Int J Neurosci. 1992. PMID: 1305637 Review.
Cited by
-
An Effective and Safe Enkephalin Analog for Antinociception.Pharmaceutics. 2021 Jun 22;13(7):927. doi: 10.3390/pharmaceutics13070927. Pharmaceutics. 2021. PMID: 34206631 Free PMC article.
References
-
- Hruby V. J., Kazmierski W., Kawasaki A. M., and Matsunaga T. O. (1991) Synthetic Chemistry and the Design of Peptide-Based Drugs, in Peptide Pharmaceuticals (Ward D. J., Ed.) Chapter 5, pp 135−184, Open University Press, Buckingham, U.K.
-
- Knapp R. J., Davis T. P., Burks T. F., and Yamamura H. I. (1991) In Peptide Pharmaceuticals, ed. (Ward D. J., Ed.) Chapter 7, pp 210−242, Open University Press, Buckingham, U.K.
-
- Partridge W. M. (1991) Peptide Drug Delivery to the Brain, Raven Press, New York.
-
- Witt K. A.; Gillespie T. J.; Huber J. D.; Egleton R. D.; Davis T. P. (2001) peptide drug modifications to enhance bioavailablility and blood-brain-barrier permeability. Peptides 22, 2329–2343. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

