Structure-activity relationships in peptide modulators of β-amyloid protein aggregation: variation in α,α-disubstitution results in altered aggregate size and morphology
- PMID: 22778850
- PMCID: PMC3368689
- DOI: 10.1021/cn100045q
Structure-activity relationships in peptide modulators of β-amyloid protein aggregation: variation in α,α-disubstitution results in altered aggregate size and morphology
Abstract
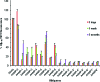
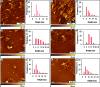
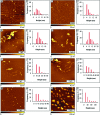
Neuronal cytotoxicity observed in Alzheimer's disease (AD) is linked to the aggregation of β-amyloid peptide (Aβ) into toxic forms. Increasing evidence points to oligomeric materials as the neurotoxic species, not Aβ fibrils; disruption or inhibition of Aβ self-assembly into oligomeric or fibrillar forms remains a viable therapeutic strategy to reduce Aβ neurotoxicity. We describe the synthesis and characterization of amyloid aggregation mitigating peptides (AAMPs) whose structure is based on the Aβ "hydrophobic core" Aβ(17-20), with α,α-disubstituted amino acids (ααAAs) added into this core as potential disrupting agents of fibril self-assembly. The number, positional distribution, and side-chain functionality of ααAAs incorporated into the AAMP sequence were found to influence the resultant aggregate morphology as indicated by ex situ experiments using atomic force microscopy (AFM) and transmission electron microscopy (TEM). For instance, AAMP-5, incorporating a sterically hindered ααAA with a diisobutyl side chain in the core sequence, disrupted Aβ(1-40) fibril formation. However, AAMP-6, with a less sterically hindered ααAA with a dipropyl side chain, altered fibril morphology, producing shorter and larger sized fibrils (compared with those of Aβ(1-40)). Remarkably, ααAA-AAMPs caused disassembly of existing Aβ fibrils to produce either spherical aggregates or protofibrillar structures, suggesting the existence of equilibrium between fibrils and prefibrillar structures.
Keywords: Alzheimer’s disease; amyloid aggregation mitigating peptides; fibrils; spherical aggregates; α,α-disubstituted amino acids; β-Amyloid.
Figures
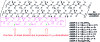




Similar articles
-
Effects of peptides derived from terminal modifications of the aβ central hydrophobic core on aβ fibrillization.ACS Chem Neurosci. 2010 Oct 20;1(10):661-78. doi: 10.1021/cn900019r. Epub 2010 Aug 26. ACS Chem Neurosci. 2010. PMID: 22778807 Free PMC article.
-
Electrochemistry of Alzheimer Disease Amyloid Beta Peptides.Curr Med Chem. 2018;25(33):4066-4083. doi: 10.2174/0929867325666180214112536. Curr Med Chem. 2018. PMID: 29446720 Review.
-
Aggregation and fibril morphology of the Arctic mutation of Alzheimer's Aβ peptide by CD, TEM, STEM and in situ AFM.J Struct Biol. 2012 Oct;180(1):174-89. doi: 10.1016/j.jsb.2012.06.010. Epub 2012 Jun 28. J Struct Biol. 2012. PMID: 22750418 Free PMC article.
-
Structural, morphological, and kinetic studies of β-amyloid peptide aggregation on self-assembled monolayers.Phys Chem Chem Phys. 2011 Sep 7;13(33):15200-10. doi: 10.1039/c1cp21156k. Epub 2011 Jul 19. Phys Chem Chem Phys. 2011. PMID: 21769359
-
Modulation of Amyloid β-Protein (Aβ) Assembly by Homologous C-Terminal Fragments as a Strategy for Inhibiting Aβ Toxicity.ACS Chem Neurosci. 2016 Jul 20;7(7):845-56. doi: 10.1021/acschemneuro.6b00154. Epub 2016 Jul 5. ACS Chem Neurosci. 2016. PMID: 27322435 Free PMC article. Review.
Cited by
-
Influence of the physiochemical properties of superparamagnetic iron oxide nanoparticles on amyloid β protein fibrillation in solution.ACS Chem Neurosci. 2013 Mar 20;4(3):475-85. doi: 10.1021/cn300196n. Epub 2013 Jan 23. ACS Chem Neurosci. 2013. PMID: 23509983 Free PMC article.
-
Effects of peptides derived from terminal modifications of the aβ central hydrophobic core on aβ fibrillization.ACS Chem Neurosci. 2010 Oct 20;1(10):661-78. doi: 10.1021/cn900019r. Epub 2010 Aug 26. ACS Chem Neurosci. 2010. PMID: 22778807 Free PMC article.
-
Cyclodextrins-Peptides/Proteins Conjugates: Synthesis, Properties and Applications.Polymers (Basel). 2021 May 27;13(11):1759. doi: 10.3390/polym13111759. Polymers (Basel). 2021. PMID: 34072062 Free PMC article. Review.
-
Polyphenols as therapeutic molecules in Alzheimer's disease through modulating amyloid pathways.Mol Neurobiol. 2015 Apr;51(2):466-79. doi: 10.1007/s12035-014-8722-9. Epub 2014 May 15. Mol Neurobiol. 2015. PMID: 24826916 Review.
-
Advancements in cyclodextrin-based controlled drug delivery: Insights into pharmacokinetic and pharmacodynamic profiles.Heliyon. 2024 Oct 30;10(21):e39917. doi: 10.1016/j.heliyon.2024.e39917. eCollection 2024 Nov 15. Heliyon. 2024. PMID: 39553547 Free PMC article. Review.
References
-
- Selkoe D. J. (1991) The molecular pathology of Alzheimers-Disease. Neuron 6, 487–498. - PubMed
-
- Hardy J.; Selkoe D. J. (2002) Medicine - The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science 297, 353–356. - PubMed
-
- Selkoe D. J. (2002) Alzheimer’s disease is a synaptic failure. Science 298, 789–791. - PubMed
-
- Sengupta P.; Garai K.; Sahoo B.; Shi Y.; Callaway D. J. E.; Maiti S. (2003) The amyloid β peptide (Aβ1−40) is thermodynamically soluble at physiological concentrations. Biochemistry 42, 10506–10513. - PubMed
-
- Bieschke J.; Zhang Q. H.; Powers E. T.; Lerner R. A.; Kelly J. W. (2005) Oxidative metabolites accelerate Alzheimer’s amyloidogenesis by a two-step mechanism, eliminating the requirement for nucleation. Biochemistry 44, 4977–4983. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

