Medicinal chemistry of competitive kainate receptor antagonists
- PMID: 22778857
- PMCID: PMC3369727
- DOI: 10.1021/cn1001039
Medicinal chemistry of competitive kainate receptor antagonists
Abstract
Kainic acid (KA) receptors belong to the group of ionotropic glutamate receptors and are expressed throughout in the central nervous system (CNS). The KA receptors have been shown to be involved in neurophysiological functions such as mossy fiber long-term potentiation (LTP) and synaptic plasticity and are thus potential therapeutic targets in CNS diseases such as schizophrenia, major depression, neuropathic pain and epilepsy. Extensive effort has been made to develop subtype-selective KA receptor antagonists in order to elucidate the physiological function of each of the five subunits known (GluK1-5). However, to date only selective antagonists for the GluK1 subunit have been discovered, which underlines the strong need for continued research in this area. The present review describes the structure-activity relationship and pharmacological profile for 10 chemically distinct classes of KA receptor antagonists comprising, in all, 45 compounds. To the medicinal chemist this information will serve as reference guidance as well as an inspiration for future effort in this field.
Keywords: Glutamate receptors; competitive antagonists; kainic acid receptors; medicinal chemistry; structure−activity relationship studies.
Figures

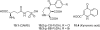
Similar articles
-
Kainate Receptor Antagonists: Recent Advances and Therapeutic Perspective.Int J Mol Sci. 2023 Jan 18;24(3):1908. doi: 10.3390/ijms24031908. Int J Mol Sci. 2023. PMID: 36768227 Free PMC article. Review.
-
N-(7-(1H-Imidazol-1-yl)-2,3-dioxo-6-(trifluoromethyl)-3,4-dihydroquinoxalin-1(2H)-yl)benzamide, a New Kainate Receptor Selective Antagonist and Analgesic: Synthesis, X-ray Crystallography, Structure-Affinity Relationships, and in Vitro and in Vivo Pharmacology.ACS Chem Neurosci. 2019 Nov 20;10(11):4685-4695. doi: 10.1021/acschemneuro.9b00479. Epub 2019 Oct 31. ACS Chem Neurosci. 2019. PMID: 31622082
-
N1-Substituted Quinoxaline-2,3-diones as Kainate Receptor Antagonists: X-ray Crystallography, Structure-Affinity Relationships, and in Vitro Pharmacology.ACS Chem Neurosci. 2019 Mar 20;10(3):1841-1853. doi: 10.1021/acschemneuro.8b00726. Epub 2019 Jan 23. ACS Chem Neurosci. 2019. PMID: 30620174
-
Mapping the ligand binding sites of kainate receptors: molecular determinants of subunit-selective binding of the antagonist [3H]UBP310.Mol Pharmacol. 2010 Dec;78(6):1036-45. doi: 10.1124/mol.110.067934. Epub 2010 Sep 13. Mol Pharmacol. 2010. PMID: 20837679 Free PMC article.
-
Lessons from crystal structures of kainate receptors.Neuropharmacology. 2017 Jan;112(Pt A):16-28. doi: 10.1016/j.neuropharm.2016.05.014. Epub 2016 May 26. Neuropharmacology. 2017. PMID: 27236079 Review.
Cited by
-
Kainate Receptors Play a Role in Modulating Synaptic Transmission in the Olfactory Bulb.Neuroscience. 2018 Nov 1;391:25-49. doi: 10.1016/j.neuroscience.2018.09.002. Epub 2018 Sep 11. Neuroscience. 2018. PMID: 30213766 Free PMC article.
-
The emerging role of kainate receptor functional dysregulation in pain.Mol Pain. 2021 Jan-Dec;17:1744806921990944. doi: 10.1177/1744806921990944. Mol Pain. 2021. PMID: 33567997 Free PMC article. Review.
-
Experimental and Computational Structural Studies of 2,3,5-Trisubstituted and 1,2,3,5-Tetrasubstituted Indoles as Non-Competitive Antagonists of GluK1/GluK2 Receptors.Molecules. 2022 Apr 12;27(8):2479. doi: 10.3390/molecules27082479. Molecules. 2022. PMID: 35458681 Free PMC article.
-
Design and Synthesis of 2,3- trans-Proline Analogues as Ligands for Ionotropic Glutamate Receptors and Excitatory Amino Acid Transporters.ACS Chem Neurosci. 2019 Jun 19;10(6):2989-3007. doi: 10.1021/acschemneuro.9b00205. Epub 2019 May 24. ACS Chem Neurosci. 2019. PMID: 31124660 Free PMC article.
-
Kainate Receptor Antagonists: Recent Advances and Therapeutic Perspective.Int J Mol Sci. 2023 Jan 18;24(3):1908. doi: 10.3390/ijms24031908. Int J Mol Sci. 2023. PMID: 36768227 Free PMC article. Review.
References
-
- Watkins J. C.; Evans R. H. (1981) Excitatory amino-acid transmitters. Annu. Rev. Pharmacool. Toxicol. 21, 165–204. - PubMed
-
- Lodge D. (2009) The history of the pharmacology and cloning of ionotropic glutamate receptors and the development of idiosyncratic nomenclature. Neuropharmacology 56, 6–21. - PubMed
-
- Bunch L.; Krogsgaard-Larsen P. (2009) Subtype Selective Kainic Acid Receptor Agonists: Discovery and Approaches to Rational Design. Med. Res. Rev. 29, 3–28. - PubMed
-
- Mellor J. R. (2006) Synaptic plasticity of kainate receptors. Biochem. Soc. Trans. 34, 949–951. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

