Switch from ER-mitochondrial to SR-mitochondrial calcium coupling during muscle differentiation
- PMID: 22784666
- PMCID: PMC3607359
- DOI: 10.1016/j.ceca.2012.05.012
Switch from ER-mitochondrial to SR-mitochondrial calcium coupling during muscle differentiation
Abstract
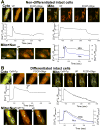
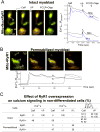
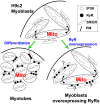
Emerging evidence indicates that mitochondria are locally coupled to endoplasmic reticulum (ER) Ca2+ release in myoblasts and to sarcoplasmic reticulum (SR) Ca2+ release in differentiated muscle fibers in order to regulate cytoplasmic calcium dynamics and match metabolism with cell activity. However, the mechanism of the developmental transition from ER to SR coupling remains unclear. We have studied mitochondrial sensing of IP3 receptor (IP3R)- and ryanodine receptor (RyR)-mediated Ca2+ signals in H9c2 myoblasts and differentiating myotubes, as well as the attendant changes in mitochondrial morphology. Mitochondria in myoblasts were largely elongated, luminally connected and relatively few in number, whereas the myotubes were densely packed with globular mitochondria that displayed limited luminal continuity. Vasopressin, an IP3-linked agonist, evoked a large cytoplasmic Ca2+ ([Ca2+]c) increase in myoblasts, whereas it elicited a smaller response in myotubes. Conversely, RyR-mediated Ca2+ release induced by caffeine, was not observed in myoblasts, but triggered a large [Ca2+]c signal in myotubes. Both the IP3R and the RyR-mediated [Ca2+]c rise was closely associated with a mitochondrial matrix Ca2+ ([Ca2+]m) signal. Every myotube that showed a [Ca2+]c spike also displayed a [Ca2+]m response. Addition of IP3 to permeabilized myoblasts and caffeine to permeabilized myotubes also resulted in a rapid [Ca2+]m rise, indicating that Ca2+ was delivered via local coupling of the ER/SR and mitochondria. Thus, as RyRs are expressed during muscle differentiation, the local connection between RyR and mitochondrial Ca2+ uptake sites also appears. When RyR1 was exogenously introduced to myoblasts by overexpression, the [Ca2+]m signal appeared together with the [Ca2+]c signal, however the mitochondrial morphology remained unchanged. Thus, RyR expression alone is sufficient to induce the steps essential for their alignment with mitochondrial Ca2+ uptake sites, whereas the mitochondrial proliferation and reshaping utilize either downstream or alternative pathways.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures



References
-
- Rusinol AE, Cui Z, Chen MH, Vance JE. A unique mitochondria-associated membrane fraction from rat liver has a high capacity for lipid synthesis and contains pre-Golgi secretory proteins including nascent lipoproteins. J Biol Chem. 1994;269:27494–502. - PubMed
-
- Rizzuto R, Pinton P, Carrington W, Fay FS, Fogarty KE, Lifshitz LM, Tuft RA, Pozzan T. Close contacts with the endoplasmic reticulum as determinants of mitochondrial Ca2+ responses. Science. 1998;280:1763–6. - PubMed
-
- de Brito OM, Scorrano L. Mitofusin 2 tethers endoplasmic reticulum to mitochondria. Nature. 2008;456:605–10. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

