Cost-effectiveness of latent tuberculosis screening before steroid therapy for idiopathic nephrotic syndrome in children
- PMID: 22784996
- PMCID: PMC3827973
- DOI: 10.1053/j.ajkd.2012.06.004
Cost-effectiveness of latent tuberculosis screening before steroid therapy for idiopathic nephrotic syndrome in children
Abstract
Background: Guidelines differ on screening recommendations for latent tuberculosis infection (LTBI) prior to immunosuppressive therapy. We aimed to determine the most cost-effective LTBI screening strategy before long-term steroid therapy in a child with new-onset idiopathic nephrotic syndrome.
Study design: Markov state-transition model.
Setting & population: 5-year-old boy with new-onset idiopathic nephrotic syndrome.
Model, perspective, & timeframe: The Markov model took a societal perspective over a lifetime horizon.
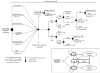
Intervention: 3 strategies were compared: universal tuberculin skin testing (TST), targeted screening using a risk-factor questionnaire, and no screening. A secondary model included the newer interferon γ release assays (IGRAs), requiring only one visit and having greater specificity than TST.
Outcomes: Marginal cost-effectiveness ratios (2010 US dollars) with effectiveness measured as quality-adjusted life-years (QALYs).
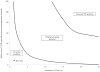
Results: At an LTBI prevalence of 1.1% (the average US childhood prevalence in our base case), a no-screening strategy dominated ($2,201; 29.3356 QALYs) targeted screening ($2,218; 29.3356 QALYs) and universal TST ($2,481; 29.3347 QALYs). At a prevalence >10.3%, targeted screening with a risk-factor questionnaire was the most cost-effective option. Higher than a prevalence of 58.5%, universal TST was preferred. In the secondary model, targeted screening with a questionnaire followed by IGRA testing was cost-effective compared with no screening in the base case when the LTBI prevalence was >4.9%.
Limitations: There is no established gold standard for the diagnosis of LTBI. Results of any modeling task are limited by the accuracy of available data.
Conclusions: Prior to starting steroid therapy, only patients in areas with a high prevalence of LTBI will benefit from universal TST. As more evidence becomes available about the use of IGRA testing in children, the assay may become a component of cost-effective screening protocols in populations with a higher burden of LTBI.
Copyright © 2012 National Kidney Foundation, Inc. Published by Elsevier Inc. All rights reserved.
Figures

Comment in
-
Screening strategies for tuberculosis in children with kidney disease: what is cost-effective?Am J Kidney Dis. 2013 Jan;61(1):3-5. doi: 10.1053/j.ajkd.2012.10.003. Am J Kidney Dis. 2013. PMID: 23245736 No abstract available.
Similar articles
-
Priorities for screening and treatment of latent tuberculosis infection in the United States.Am J Respir Crit Care Med. 2011 Sep 1;184(5):590-601. doi: 10.1164/rccm.201101-0181OC. Am J Respir Crit Care Med. 2011. PMID: 21562129 Free PMC article.
-
Cost-effectiveness of Testing and Treatment for Latent Tuberculosis Infection in Residents Born Outside the United States With and Without Medical Comorbidities in a Simulation Model.JAMA Intern Med. 2017 Dec 1;177(12):1755-1764. doi: 10.1001/jamainternmed.2017.3941. JAMA Intern Med. 2017. PMID: 29049814 Free PMC article.
-
Comparing the cost-effectiveness of two screening strategies for latent tuberculosis infection in Portugal.Pulmonology. 2021 Nov-Dec;27(6):493-499. doi: 10.1016/j.pulmoe.2021.04.002. Epub 2021 May 27. Pulmonology. 2021. PMID: 34053903 Review.
-
Economic evaluation of diagnosis and treatment for latent tuberculosis infection among contacts of pulmonary tuberculosis patients in Thailand.Sci Rep. 2024 Jul 31;14(1):17693. doi: 10.1038/s41598-024-68452-1. Sci Rep. 2024. PMID: 39085338 Free PMC article.
-
Tuberculin skin test - Outdated or still useful for Latent TB infection screening?Int J Infect Dis. 2019 Mar;80S:S20-S22. doi: 10.1016/j.ijid.2019.01.048. Epub 2019 Feb 6. Int J Infect Dis. 2019. PMID: 30738186 Review.
Cited by
-
Tuberculosis in Takayasu arteritis: a retrospective study in 1105 Chinese patients.J Geriatr Cardiol. 2019 Aug;16(8):648-655. doi: 10.11909/j.issn.1671-5411.2019.08.003. J Geriatr Cardiol. 2019. PMID: 31555333 Free PMC article.
-
Scoping Review of Economic Analyses of Rare Kidney Diseases.Kidney Int Rep. 2024 Sep 12;9(12):3553-3569. doi: 10.1016/j.ekir.2024.09.004. eCollection 2024 Dec. Kidney Int Rep. 2024. PMID: 39698356 Free PMC article.
-
Economic evaluation of novel Mycobacterium tuberculosis specific antigen-based skin tests for detection of TB infection: A modelling study.PLOS Glob Public Health. 2023 Dec 20;3(12):e0002573. doi: 10.1371/journal.pgph.0002573. eCollection 2023. PLOS Glob Public Health. 2023. PMID: 38117825 Free PMC article.
-
Cost Effectiveness of Preventive Treatment for Tuberculosis in Special High-Risk Populations.Pharmacoeconomics. 2015 Aug;33(8):783-809. doi: 10.1007/s40273-015-0267-x. Pharmacoeconomics. 2015. PMID: 25774015
-
US-Based Drug Cost Parameter Estimation for Economic Evaluations.Med Decis Making. 2015 Jul;35(5):622-32. doi: 10.1177/0272989X14563987. Epub 2014 Dec 22. Med Decis Making. 2015. PMID: 25532826 Free PMC article.
References
-
- American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America: Controlling Tuberculosis in the United States. Am J Respir Crit Care Med. 2005;172(9):1169–1226. - PubMed
-
- World Health Organization. [Accessed December 12, 2011.];Global Tuberculosis Control. 2011 http://www.who.int/tb/publications/global_report/2011/gtbr11_full.pdf.
-
- Cruz AT, Geltemeyer AM, Starke JR, Flores JA, Graviss EA, Smith KC. Comparing the tuberculin skin test and T-SPOT.TB blood test in children. Pediatrics. 2011;127(1):e31–38. - PubMed
-
- Horsburgh CR, Jr, Goldberg S, Bethel J, et al. Latent TB infection treatment acceptance and completion in the United States and Canada. Chest. 2010;137(2):401–409. - PubMed
-
- Mazurek GH, Jereb J, Vernon A, LoBue P, Goldberg S, Castro K. Updated guidelines for using Interferon Gamma Release Assays to detect Mycobacterium tuberculosis infection - United States, 2010. MMWR Recomm Rep. 2010;59(RR-5):1–25. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

